Academic Clinical Trials Submitted to an Institutional Ethics Committee at a Tertiary Care Center: A Retrospective Analysis
- PMID: 38440003
- PMCID: PMC10909758
- DOI: 10.7759/cureus.53476
Academic Clinical Trials Submitted to an Institutional Ethics Committee at a Tertiary Care Center: A Retrospective Analysis
Abstract
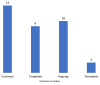
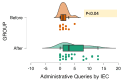
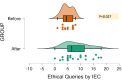
Background The role of academia in clinical research has given rise to the concept of academic clinical trials (ACTs), which are vital in generating evidence. Through the implementation of the New Drugs and Clinical Trials Rules-2019 (NDCTR-2019) rules, the Institutional Ethics Committee (IEC) has obtained a quasi-regulatory role. The study aims to assess the challenges the IEC faced when processing, approving, and monitoring ACTs. The other objectives included the number of ACTs submitted to the IEC, as well as administrative, scientific, and ethical issues stated by the IEC and the Drug Controller General of India (DCGI) authorities. We also aimed to provide some insight into the type of decision made by IEC and DCGI - the delay or inconsistency between the queries. Methods This retrospective study was conducted in the IEC of a tertiary care hospital, Mumbai, Maharashtra, India. A comprehensive search of the IEC database was carried out by the study team, and only those protocols of ACTs submitted to IEC between January 2015 and December 2021 were included. The studies submitted between January 2015 and February 2019, i.e., before the release of NDCTR-2019, were classified as the "Before" category. All subsequently submitted protocols were grouped together as the "After" group. Descriptive statistics were used to represent the data, while comparison between the two timeframes were made using the Mann-Whitney U test with a level of significance at 5%. Results This six-year study showed that merely 1.4% (34/2400) trials fulfilled the criteria of an ACT. An increase in the ACT protocol submission was noted in the "After" group (20 vs. 14). Most ACTs were drug trials, with 67.6% (23/34) trials conducted majorly in the Department of Anesthesiology. There was a statistical increase in time query reply by the principal investigator to IEC and the time between submission and approval in the "After" group (p<0.05). IEC sent out 94 administrative, 565 scientific, and 216 ethical queries. On IEC monitoring, protocol deviations were noted; nonetheless, no ACTs reported protocol deviations or serious adverse events. Conclusions Since the implementation of NDCTR-2019, IEC has taken on a quasi-regulatory function, and there has been an increase in the caliber of IEC monitoring and adherence to ethical norms.
Keywords: academic clinical trials; central licensing authority; clinical trials; drugs controller general of india; investigator-initiated studies; new drugs and clinical trials rules 2019.
Copyright © 2024, Shetty et al.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

Similar articles
-
Audit of principal investigator's compliance for submission of continue review application and decisions taken on lapses in validity of approval by the Institutional Ethics Committee at tertiary oncology center in Navi Mumbai.Perspect Clin Res. 2022 Jul-Sep;13(3):145-150. doi: 10.4103/picr.PICR_102_20. Epub 2021 Feb 8. Perspect Clin Res. 2022. PMID: 35928644 Free PMC article.
-
Investigator knowledge, awareness, and registrations of academic clinical trials with the Clinical Trial Registry of India: An observational study.Perspect Clin Res. 2023 Oct-Dec;14(4):187-193. doi: 10.4103/picr.picr_68_23. Epub 2023 Aug 8. Perspect Clin Res. 2023. PMID: 38025286 Free PMC article.
-
Projects not initiated by investigators: a retrospective analysis of the queries raised by the institutional ethics committees of a teaching hospital.J Postgrad Med. 2014 Jan-Mar;60(1):46-50. doi: 10.4103/0022-3859.128808. J Postgrad Med. 2014. PMID: 24625939
-
Influenza virus vaccine live intranasal--MedImmune vaccines: CAIV-T, influenza vaccine live intranasal.Drugs R D. 2003;4(5):312-9. doi: 10.2165/00126839-200304050-00007. Drugs R D. 2003. PMID: 12952502 Review.
-
Oblimersen: Augmerosen, BCL-2 antisense oligonucleotide - Genta, G 3139, GC 3139, oblimersen sodium.Drugs R D. 2007;8(5):321-34. doi: 10.2165/00126839-200708050-00006. Drugs R D. 2007. PMID: 17767397 Review.
References
-
- The significance of clinical trials. Novitzke JM. https://pubmed.ncbi.nlm.nih.gov/22518214/ J Vasc Interv Neurol. 2008;1:31. - PMC - PubMed
-
- Strengths and limitations of industry vs. academic randomized controlled trials. Laterre PF, François B. Clin Microbiol Infect. 2015;21:906–909. - PubMed
-
- Investigator Initiated Trials (IITs): Introduction, regulatory view, study design preferences, and publication strategies. [ Sep; 2023 ];https://prorelixresearch.com/investigator-initiated-trials-iits-introduc... ProRelix Research. 2023 8
LinkOut - more resources
Full Text Sources
