Selective Quantification of Bacteria in Mixtures by Using Glycosylated Polypyrrole/Hydrogel Nanolayers
- PMID: 38442898
- PMCID: PMC10959108
- DOI: 10.1021/acsami.3c14387
Selective Quantification of Bacteria in Mixtures by Using Glycosylated Polypyrrole/Hydrogel Nanolayers
Abstract
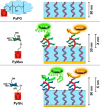
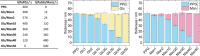
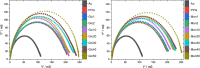
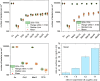
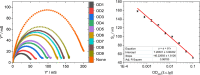
Here, we present a covalent nanolayer system that consists of a conductive and biorepulsive base layer topped by a layer carrying biorecognition sites. The layers are built up by electropolymerization of pyrrole derivatives that either carry polyglycerol brushes (for biorepulsivity) or glycoside moieties (as biorecognition sites). The polypyrrole backbone makes the resulting nanolayer systems conductive, opening the opportunity for constructing an electrochemistry-based sensor system. The basic concept of the sensor exploits the highly selective binding of carbohydrates by certain harmful bacteria, as bacterial adhesion and infection are a major threat to human health, and thus, a sensitive and selective detection of the respective bacteria by portable devices is highly desirable. To demonstrate the selectivity, two strains of Escherichia coli were selected. The first strain carries type 1 fimbriae, terminated by a lectin called FimH, which recognizes α-d-mannopyranosides, which is a carbohydrate that is commonly found on endothelial cells. The otherE. coli strain was of a strain that lacked this particular lectin. It could be demonstrated that hybrid nanolayer systems containing a very thin carbohydrate top layer (2 nm) show the highest discrimination (factor 80) between the different strains. Using electrochemical impedance spectroscopy, it was possible to quantify in vivo the type 1-fimbriated E. coli down to an optical density of OD600 = 0.0004 with a theoretical limit of 0.00005. Surprisingly, the selectivity and sensitivity of the sensing remained the same even in the presence of a large excess of nonbinding bacteria, making the system useful for the rapid and selective detection of pathogens in complex matrices. As the presented covalent nanolayer system is modularly built, it opens the opportunity to develop a broad band of mobile sensing devices suitable for various field applications such as bedside diagnostics or monitoring for bacterial contamination, e.g., in bioreactors.
Keywords: biorepulsive; distinction of bacteria; electrochemical sensor; glycosides; nanolayer polymerization; polypyrrole.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

