Role of biophysics and mechanobiology in podocyte physiology
- PMID: 38443711
- PMCID: PMC12103212
- DOI: 10.1038/s41581-024-00815-3
Role of biophysics and mechanobiology in podocyte physiology
Abstract
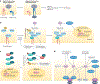
Podocytes form the backbone of the glomerular filtration barrier and are exposed to various mechanical forces throughout the lifetime of an individual. The highly dynamic biomechanical environment of the glomerular capillaries greatly influences the cell biology of podocytes and their pathophysiology. Throughout the past two decades, a holistic picture of podocyte cell biology has emerged, highlighting mechanobiological signalling pathways, cytoskeletal dynamics and cellular adhesion as key determinants of biomechanical resilience in podocytes. This biomechanical resilience is essential for the physiological function of podocytes, including the formation and maintenance of the glomerular filtration barrier. Podocytes integrate diverse biomechanical stimuli from their environment and adapt their biophysical properties accordingly. However, perturbations in biomechanical cues or the underlying podocyte mechanobiology can lead to glomerular dysfunction with severe clinical consequences, including proteinuria and glomerulosclerosis. As our mechanistic understanding of podocyte mechanobiology and its role in the pathogenesis of glomerular disease increases, new targets for podocyte-specific therapeutics will emerge. Treating glomerular diseases by targeting podocyte mechanobiology might improve therapeutic precision and efficacy, with potential to reduce the burden of chronic kidney disease on individuals and health-care systems alike.
© 2024. Springer Nature Limited.
Conflict of interest statement
Competing Interests
The authors declare no competing interests.
Figures

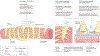

References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

