A real-world study of treatment patterns following disease progression in epithelial ovarian cancer patients undergoing poly-ADP-ribose polymerase inhibitor maintenance therapy
- PMID: 38444005
- PMCID: PMC10913203
- DOI: 10.1186/s13048-024-01381-9
A real-world study of treatment patterns following disease progression in epithelial ovarian cancer patients undergoing poly-ADP-ribose polymerase inhibitor maintenance therapy
Abstract
Background: The efficacy of subsequent therapy after poly-ADP-ribose polymerase (PARP) inhibitor maintenance treatment has raised concerns. Retrospective studies show worse outcomes for platinum-based chemotherapy after progression of PARP inhibitor-maintenance therapy, especially in BRCA-mutant patients. We aimed to describe subsequent therapy in ovarian cancer patients after PARP inhibitor-maintenance therapy and evaluate their response to treatment. We focused on chemotherapy for patients with a progression-free interval (PFI) of ≥ 6 months after prior platinum treatment, based on BRCA status.
Methods: We analyzed real-world data from Peking University Cancer Hospital, subsequent therapy after progression to PARP inhibitor-maintenance therapy for epithelial ovarian cancer between January 2016 and December 2022. Clinicopathological characteristics and treatment outcomes were extracted from medical records. The last follow-up was in May 2023.
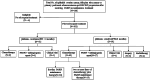
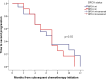
Results: A total of 102 patients were included, of which 29 (28.4%) had a germline BRCA1/2 mutation and 73 (71.6%) exhibited BRCA1/2 wild-type mutations. The PARP inhibitors used were Olaparib (n = 62, 60.8%), Niraparib (n = 35, 34.3%), and others (n = 5, 4.9%). The overall response rate (ORR) was 41.2%, and the median time to second progression (mTTSP) was 8.1 months (95%CI 5.8-10.2). Of 91 platinum-sensitive patients (PFI ≥ 6 months) after progression to PARP inhibitor-maintenance therapy, 65 patients subsequently received platinum regimens. Among them, 30 had received one line of chemotherapy before PARP inhibitor-maintenance therapy. Analysis of these 30 patients by BRCA status showed an ORR of 16.7% versus 33.3% and mTTSP of 7.1 (95% CI 4.9-9.1) versus 6.2 months (95% CI 3.7-8.3, P = 0.550), for BRCA-mutant and wild-type patients, respectively. For the remaining 35 patients who had received two or more lines of chemotherapy before PARP inhibitor-maintenance therapy, ORR was 57.1% versus 42.9%, and mTTSP was 18.0 (95% CI 5.0-31.0) versus 8.0 months (95% CI 4.9-11.1, P = 0.199), for BRCA-mutant and wild-type patients, respectively.
Conclusion: No differences in survival outcomes were observed among patients with different BRCA statuses. Furthermore, for patients who had undergone two or more lines of chemotherapy before PARP inhibitor maintenance therapy, no negative effects of PARP inhibitors on subsequent treatment were found, regardless of BRCA status.
Keywords: BRCA; Epithelial ovarian cancer; Maintenance therapy; PARP; Subsequent therapy.
© 2024. The Author(s).
Conflict of interest statement
The authors have no relevant financial or non-financial interests to disclose
Figures

Similar articles
-
Efficacy of subsequent chemotherapy for patients with BRCA1/2-mutated recurrent epithelial ovarian cancer progressing on olaparib versus placebo maintenance: post-hoc analyses of the SOLO2/ENGOT Ov-21 trial.Ann Oncol. 2022 Oct;33(10):1021-1028. doi: 10.1016/j.annonc.2022.06.011. Epub 2022 Jun 27. Ann Oncol. 2022. PMID: 35772665 Clinical Trial.
-
Rucaparib in relapsed, platinum-sensitive high-grade ovarian carcinoma (ARIEL2 Part 1): an international, multicentre, open-label, phase 2 trial.Lancet Oncol. 2017 Jan;18(1):75-87. doi: 10.1016/S1470-2045(16)30559-9. Epub 2016 Nov 29. Lancet Oncol. 2017. PMID: 27908594 Clinical Trial.
-
Maintenance olaparib rechallenge in patients with platinum-sensitive relapsed ovarian cancer previously treated with a PARP inhibitor (OReO/ENGOT-ov38): a phase IIIb trial.Ann Oncol. 2023 Dec;34(12):1152-1164. doi: 10.1016/j.annonc.2023.09.3110. Epub 2023 Oct 4. Ann Oncol. 2023. PMID: 37797734 Clinical Trial.
-
Current strategies for the targeted treatment of high-grade serous epithelial ovarian cancer and relevance of BRCA mutational status.J Ovarian Res. 2019 Jan 28;12(1):9. doi: 10.1186/s13048-019-0484-6. J Ovarian Res. 2019. PMID: 30691488 Free PMC article. Review.
-
Using PARP Inhibitors in the Treatment of Patients With Ovarian Cancer.Curr Treat Options Oncol. 2018 Nov 15;19(12):1. doi: 10.1007/s11864-018-0572-7. Curr Treat Options Oncol. 2018. PMID: 30535808 Free PMC article. Review.
Cited by
-
Prevalence of BRCA1 and BRCA2 mutations in ovarian cancer patients from Yunnan Province in southwest China.Eur J Cancer Prev. 2025 May 1;34(3):231-240. doi: 10.1097/CEJ.0000000000000931. Epub 2024 Oct 25. Eur J Cancer Prev. 2025. PMID: 39513675 Free PMC article.
-
Precision medicine in gynecological cancer (Review).Biomed Rep. 2025 Jan 8;22(3):43. doi: 10.3892/br.2025.1921. eCollection 2025 Mar. Biomed Rep. 2025. PMID: 39810899 Free PMC article. Review.
References
-
- Pujade-Lauraine E, Ledermann JA, Selle F, Gebski V, Penson RT, Oza AM, et al. Olaparib tablets as maintenance therapy in patients with platinum-sensitive, relapsed ovarian cancer and a BRCA1/2 mutation (SOLO2/ENGOT-Ov21): a double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Oncol. 2017;18(9):1274–84. doi: 10.1016/s1470-2045(17)30469-2. - DOI - PubMed
-
- Coleman RL, Oza AM, Lorusso D, Aghajanian C, Oaknin A, Dean A, et al. Rucaparib maintenance treatment for recurrent ovarian carcinoma after response to platinum therapy (ARIEL3): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2017;390(10106):1949–61. doi: 10.1016/s0140-6736(17)32440-6. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

