Updating perspectives on spinal cord function: motor coordination, timing, relational processing, and memory below the brain
- PMID: 38444825
- PMCID: PMC10912355
- DOI: 10.3389/fnsys.2024.1184597
Updating perspectives on spinal cord function: motor coordination, timing, relational processing, and memory below the brain
Abstract
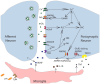
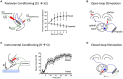
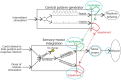
Those studying neural systems within the brain have historically assumed that lower-level processes in the spinal cord act in a mechanical manner, to relay afferent signals and execute motor commands. From this view, abstracting temporal and environmental relations is the province of the brain. Here we review work conducted over the last 50 years that challenges this perspective, demonstrating that mechanisms within the spinal cord can organize coordinated behavior (stepping), induce a lasting change in how pain (nociceptive) signals are processed, abstract stimulus-stimulus (Pavlovian) and response-outcome (instrumental) relations, and infer whether stimuli occur in a random or regular manner. The mechanisms that underlie these processes depend upon signal pathways (e.g., NMDA receptor mediated plasticity) analogous to those implicated in brain-dependent learning and memory. New data show that spinal cord injury (SCI) can enable plasticity within the spinal cord by reducing the inhibitory effect of GABA. It is suggested that the signals relayed to the brain may contain information about environmental relations and that spinal cord systems can coordinate action in response to descending signals from the brain. We further suggest that the study of stimulus processing, learning, memory, and cognitive-like processing in the spinal cord can inform our views of brain function, providing an attractive model system. Most importantly, the work has revealed new avenues of treatment for those that have suffered a SCI.
Keywords: ionic plasticity; learning; metaplasticity; pain; plasticity; recovery; spinal cord injury.
Copyright © 2024 Grau, Hudson, Johnston and Partipilo.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures




Similar articles
-
Learning to promote recovery after spinal cord injury.Exp Neurol. 2020 Aug;330:113334. doi: 10.1016/j.expneurol.2020.113334. Epub 2020 Apr 28. Exp Neurol. 2020. PMID: 32353465 Free PMC article. Review.
-
Metaplasticity within the spinal cord: Evidence brain-derived neurotrophic factor (BDNF), tumor necrosis factor (TNF), and alterations in GABA function (ionic plasticity) modulate pain and the capacity to learn.Neurobiol Learn Mem. 2018 Oct;154:121-135. doi: 10.1016/j.nlm.2018.04.007. Epub 2018 Apr 7. Neurobiol Learn Mem. 2018. PMID: 29635030 Free PMC article.
-
Behavioral studies of spinal conditioning: The spinal cord is smarter than you think it is.J Exp Psychol Anim Learn Cogn. 2022 Oct;48(4):435-457. doi: 10.1037/xan0000332. Epub 2022 Jul 28. J Exp Psychol Anim Learn Cogn. 2022. PMID: 35901417 Free PMC article.
-
Central nociceptive sensitization vs. spinal cord training: opposing forms of plasticity that dictate function after complete spinal cord injury.Front Physiol. 2012 Oct 4;3:396. doi: 10.3389/fphys.2012.00396. eCollection 2012. Front Physiol. 2012. PMID: 23060820 Free PMC article.
-
Metaplasticity and behavior: how training and inflammation affect plastic potential within the spinal cord and recovery after injury.Front Neural Circuits. 2014 Sep 8;8:100. doi: 10.3389/fncir.2014.00100. eCollection 2014. Front Neural Circuits. 2014. PMID: 25249941 Free PMC article. Review.
Cited by
-
Towards non-invasive imaging through spinal-cord generated magnetic fields.Front Med Technol. 2024 Oct 9;6:1470970. doi: 10.3389/fmedt.2024.1470970. eCollection 2024. Front Med Technol. 2024. PMID: 39445170 Free PMC article.
-
Enhancing mitophagy by ligustilide through BNIP3-LC3 interaction attenuates oxidative stress-induced neuronal apoptosis in spinal cord injury.Int J Biol Sci. 2024 Aug 12;20(11):4382-4406. doi: 10.7150/ijbs.98051. eCollection 2024. Int J Biol Sci. 2024. PMID: 39247814 Free PMC article.
References
-
- Allen C., Grau J. W., Meagher M. W. (2002). “The lower bounds of cognition: what do spinal cords reveal?” in The Oxford handbook of philosophy of neuroscience. ed. Bickle J. (Oxford: Oxford Press; ), 129–142.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

