Therapeutic role of miR-26a on cardiorenal injury in a mice model of angiotensin-II induced chronic kidney disease through inhibition of LIMS1/ILK pathway
- PMID: 38445356
- PMCID: PMC11745862
- DOI: 10.1097/CM9.0000000000002978
Therapeutic role of miR-26a on cardiorenal injury in a mice model of angiotensin-II induced chronic kidney disease through inhibition of LIMS1/ILK pathway
Abstract
Background: Chronic kidney disease (CKD) is associated with common pathophysiological processes, such as inflammation and fibrosis, in both the heart and the kidney. However, the underlying molecular mechanisms that drive these processes are not yet fully understood. Therefore, this study focused on the molecular mechanism of heart and kidney injury in CKD.
Methods: We generated an microRNA (miR)-26a knockout (KO) mouse model to investigate the role of miR-26a in angiotensin (Ang)-II-induced cardiac and renal injury. We performed Ang-II modeling in wild type (WT) mice and miR-26a KO mice, with six mice in each group. In addition, Ang-II-treated AC16 cells and HK2 cells were used as in vitro models of cardiac and renal injury in the context of CKD. Histological staining, immunohistochemistry, quantitative real-time polymerase chain reaction (PCR), and Western blotting were applied to study the regulation of miR-26a on Ang-II-induced cardiac and renal injury. Immunofluorescence reporter assays were used to detect downstream genes of miR-26a, and immunoprecipitation was employed to identify the interacting protein of LIM and senescent cell antigen-like domain 1 (LIMS1). We also used an adeno-associated virus (AAV) to supplement LIMS1 and explored the specific regulatory mechanism of miR-26a on Ang-II-induced cardiac and renal injury. Dunnett's multiple comparison and t -test were used to analyze the data.
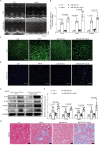
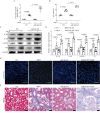
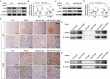
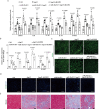
Results: Compared with the control mice, miR-26a expression was significantly downregulated in both the kidney and the heart after Ang-II infusion. Our study identified LIMS1 as a novel target gene of miR-26a in both heart and kidney tissues. Downregulation of miR-26a activated the LIMS1/integrin-linked kinase (ILK) signaling pathway in the heart and kidney, which represents a common molecular mechanism underlying inflammation and fibrosis in heart and kidney tissues during CKD. Furthermore, knockout of miR-26a worsened inflammation and fibrosis in the heart and kidney by inhibiting the LIMS1/ILK signaling pathway; on the contrary, supplementation with exogenous miR-26a reversed all these changes.
Conclusions: Our findings suggest that miR-26a could be a promising therapeutic target for the treatment of cardiorenal injury in CKD. This is attributed to its ability to regulate the LIMS1/ILK signaling pathway, which represents a common molecular mechanism in both heart and kidney tissues.
Copyright © 2024 The Chinese Medical Association, produced by Wolters Kluwer, Inc. under the CC-BY-NC-ND license.
Conflict of interest statement
None.
Figures

Similar articles
-
HIF-1α and adaptor protein LIM and senescent cell antigen-like domains protein 1 axis promotes tubulointerstitial fibrosis by interacting with vimentin in angiotensin II-induced hypertension.Br J Pharmacol. 2024 Sep;181(17):3098-3117. doi: 10.1111/bph.16358. Epub 2024 May 3. Br J Pharmacol. 2024. PMID: 38698737
-
Adiponectin attenuates kidney injury and fibrosis in deoxycorticosterone acetate-salt and angiotensin II-induced CKD mice.Am J Physiol Renal Physiol. 2018 Sep 1;315(3):F558-F571. doi: 10.1152/ajprenal.00137.2018. Epub 2018 Jun 6. Am J Physiol Renal Physiol. 2018. PMID: 29873514 Free PMC article.
-
MiR-26a-5p alleviates cardiac hypertrophy and dysfunction via targeting ADAM17.Cell Biol Int. 2021 Nov;45(11):2357-2367. doi: 10.1002/cbin.11685. Epub 2021 Aug 23. Cell Biol Int. 2021. PMID: 34370360
-
Chronic kidney disease induced by an adenine rich diet upregulates integrin linked kinase (ILK) and its depletion prevents the disease progression.Biochim Biophys Acta Mol Basis Dis. 2019 Jun 1;1865(6):1284-1297. doi: 10.1016/j.bbadis.2019.01.024. Epub 2019 Feb 3. Biochim Biophys Acta Mol Basis Dis. 2019. PMID: 30726718
-
MicroRNA-26a in respiratory diseases: mechanisms and therapeutic potential.Mol Biol Rep. 2024 May 8;51(1):627. doi: 10.1007/s11033-024-09576-5. Mol Biol Rep. 2024. PMID: 38717532 Review.
References
-
- Kalantar-Zadeh K, Jafar TH, Nitsch D, Neuen BL, Perkovic V. Chronic kidney disease. Lancet 2021;398:786–802. doi: 10.1016/S0140-6736(21)00519-5. - PubMed
-
- Ruiz-Ortega M, Rayego-Mateos S, Lamas S, Ortiz A, Rodrigues-Diez RR. Targeting the progression of chronic kidney disease. Nat Rev Nephrol 2020;16:269–288. doi: 10.1038/s41581-019-0248-y. - PubMed
-
- Treiber T, Treiber N, Meister G. Regulation of microRNA biogenesis and its crosstalk with other cellular pathways. Nat Rev Mol Cell Biol 2019;20:5–20. doi: 10.1038/s41580-018-0059-1. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

