Shotgun metagenomics and systemic targeted metabolomics highlight indole-3-propionic acid as a protective gut microbial metabolite against influenza infection
- PMID: 38445660
- PMCID: PMC10936607
- DOI: 10.1080/19490976.2024.2325067
Shotgun metagenomics and systemic targeted metabolomics highlight indole-3-propionic acid as a protective gut microbial metabolite against influenza infection
Abstract
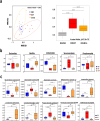
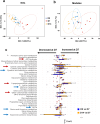
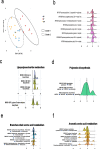
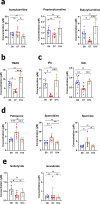
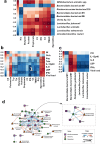
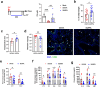
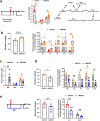
The gut-to-lung axis is critical during respiratory infections, including influenza A virus (IAV) infection. In the present study, we used high-resolution shotgun metagenomics and targeted metabolomic analysis to characterize influenza-associated changes in the composition and metabolism of the mouse gut microbiota. We observed several taxonomic-level changes on day (D)7 post-infection, including a marked reduction in the abundance of members of the Lactobacillaceae and Bifidobacteriaceae families, and an increase in the abundance of Akkermansia muciniphila. On D14, perturbation persisted in some species. Functional scale analysis of metagenomic data revealed transient changes in several metabolic pathways, particularly those leading to the production of short-chain fatty acids (SCFAs), polyamines, and tryptophan metabolites. Quantitative targeted metabolomics analysis of the serum revealed changes in specific classes of gut microbiota metabolites, including SCFAs, trimethylamine, polyamines, and indole-containing tryptophan metabolites. A marked decrease in indole-3-propionic acid (IPA) blood level was observed on D7. Changes in microbiota-associated metabolites correlated with changes in taxon abundance and disease marker levels. In particular, IPA was positively correlated with some Lactobacillaceae and Bifidobacteriaceae species (Limosilactobacillus reuteri, Lactobacillus animalis) and negatively correlated with Bacteroidales bacterium M7, viral load, and inflammation markers. IPA supplementation in diseased animals reduced viral load and lowered local (lung) and systemic inflammation. Treatment of mice with antibiotics targeting IPA-producing bacteria before infection enhanced viral load and lung inflammation, an effect inhibited by IPA supplementation. The results of this integrated metagenomic-metabolomic analysis highlighted IPA as an important contributor to influenza outcomes and a potential biomarker of disease severity.
Keywords: Influenza; disease severity; gut microbiota; indole-3-propionic acid; metabolomics; shotgun metagenomics.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as potential conflicts of interest.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
