Peroxygenase-Catalyzed Allylic Oxidation Unlocks Telescoped Synthesis of (1 S,3 R)-3-Hydroxycyclohexanecarbonitrile
- PMID: 38449536
- PMCID: PMC10913032
- DOI: 10.1021/acscatal.4c00177
Peroxygenase-Catalyzed Allylic Oxidation Unlocks Telescoped Synthesis of (1 S,3 R)-3-Hydroxycyclohexanecarbonitrile
Abstract
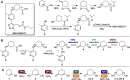
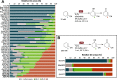
The unmatched chemo-, regio-, and stereoselectivity of enzymes renders them powerful catalysts in the synthesis of chiral active pharmaceutical ingredients (APIs). Inspired by the discovery route toward the LPA1-antagonist BMS-986278, access to the API building block (1S,3R)-3-hydroxycyclohexanecarbonitrile was envisaged using an ene reductase (ER) and alcohol dehydrogenase (ADH) to set both stereocenters. Starting from the commercially available cyclohexene-1-nitrile, a C-H oxyfunctionalization step was required to introduce the ketone functional group, yet several chemical allylic oxidation strategies proved unsuccessful. Enzymatic strategies for allylic oxidation are underdeveloped, with few examples on selected substrates with cytochrome P450s and unspecific peroxygenases (UPOs). In this case, UPOs were found to catalyze the desired allylic oxidation with high chemo- and regioselectivity, at substrate loadings of up to 200 mM, without the addition of organic cosolvents, thus enabling the subsequent ER and ADH steps in a three-step one-pot cascade. UPOs even displayed unreported enantioselective oxyfunctionalization and overoxidation of the substituted cyclohexene. After screening of enzyme panels, the final product was obtained at titers of 85% with 97% ee and 99% de, with a substrate loading of 50 mM, the ER being the limiting step. This synthetic approach provides the first example of a three-step, one-pot UPO-ER-ADH cascade and highlights the potential for UPOs to catalyze diverse enantioselective allylic hydroxylations and oxidations that are otherwise difficult to achieve.
© 2024 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



References
-
- Reß T.; Hummel W.; Hanlon S. P.; Iding H.; Gröger H. The organic-synthetic potential of recombinant ene reductases: substrate-scope evaluation and process optimization. ChemCatChem. 2015, 7, 1302–1311. 10.1002/cctc.201402903. - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous
