Toxoplasma gondii Me49 and NED strains arrest host cell cycle progression and alter chromosome segregation in a strain-independent manner
- PMID: 38450167
- PMCID: PMC10915083
- DOI: 10.3389/fmicb.2024.1336267
Toxoplasma gondii Me49 and NED strains arrest host cell cycle progression and alter chromosome segregation in a strain-independent manner
Abstract
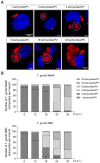
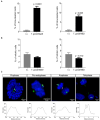
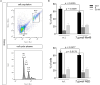
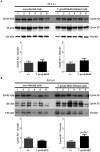
Toxoplasma gondii is an obligate intracellular parasite that modulates a broad range of host cell functions to guarantee its intracellular development and replication. T. gondii includes three classical clonal lineages exhibiting different degrees of virulence. Regarding the genetic diversity of T. gondii circulating in Europe, type II strains and, to a lesser extent, type III strains are the dominant populations, both in humans and animals. Infections with the type I strain led to widespread parasite dissemination and death in mice, while type III is considered avirulent. Previously, we demonstrated that primary endothelial cells infected with the T. gondii RH strain (haplotype I) were arrested in the G2/M-phase transition, triggering cytokinesis failure and chromosome missegregation. Since T. gondii haplotypes differ in their virulence, we here studied whether T. gondii-driven host cell cycle perturbation is strain-dependent. Primary endothelial cells were infected with T. gondii Me49 (type II strain) or NED (type III strain), and their growth kinetics were compared up to cell lysis (6-30 h p. i.). In this study, only slight differences in the onset of full proliferation were observed, and developmental data in principle matched those of the RH strain. FACS-based DNA quantification to estimate cell proportions experiencing different cell cycle phases (G0/1-, S-, and G2/M-phase) revealed that Me49 and NED strains both arrested the host cell cycle in the S-phase. Cyclins A2 and B1 as key molecules of S- and M-phase were not changed by Me49 infection, while NED infection induced cyclin B1 upregulation. To analyze parasite-driven alterations during mitosis, we demonstrated that both Me49 and NED infections led to impaired host cellular chromosome segregation and irregular centriole overduplication. Moreover, in line with the RH strain, both strains boosted the proportion of binucleated cells within infected endothelial cell layers, thereby indicating enhanced cytokinesis failure. Taken together, we demonstrate that all parasite-driven host cell cycle arrest, chromosome missegregation, and binucleated phenotypes are T. gondii-specific but strain independent.
Keywords: Me49 strain; NED strain; Toxoplasma gondii; cell cycle arrest; cell cycle dysregulation; haplotypes.
Copyright © 2024 Rojas-Barón, Hermosilla, Taubert and Velásquez.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Alberts B., Johnson A., Lewis J., Raff M., Roberts K., Walter P. (2007). “The cell cycle” in Molecular biology of the cell 5th edition, Garland Science, (New York: W.W. Norton & Company; ).
LinkOut - more resources
Full Text Sources

