Diversity, community structure, and abundance of nirS-type denitrifying bacteria on suspended particulate matter in coastal high-altitude aquaculture pond water
- PMID: 38454013
- PMCID: PMC10920899
- DOI: 10.1038/s41598-024-56196-x
Diversity, community structure, and abundance of nirS-type denitrifying bacteria on suspended particulate matter in coastal high-altitude aquaculture pond water
Abstract
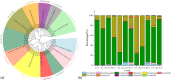
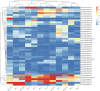
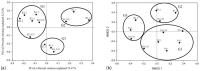
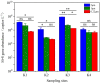
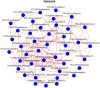
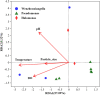
Denitrifying bacteria harboring the nitrate reductase S (nirS) gene convert active nitrogen into molecular nitrogen, and alleviate eutrophication in aquaculture water. Suspended particulate matter (SPM) is an important component of aquaculture water and a carrier for denitrification. SPM with different particle sizes were collected from a coastal high-altitude aquaculture pond in Maoming City, China. Diversity, community structure, abundance of nirS-type denitrifying bacteria on SPM and environmental influencing factors were studied using high-throughput sequencing, fluorescence quantitative PCR, and statistical analysis. Pseudomonas, Halomonas, and Wenzhouxiangella were the dominant genera of nirS-type denitrifying bacteria on SPM from the ponds. Network analysis revealed Pseudomonas and Halomonas as the key genera involved in the interaction of nirS-type denitrifying bacteria on SPM in the ponds. qPCR indicated a trend toward greater nirS gene abundance in progressively larger SPM. Dissolved oxygen, pH, temperature, and SPM particle size were the main environmental factors influencing changes in the nirS-type denitrifying bacterial community on SPM in coastal high-altitude aquaculture pond water. These findings increase our understanding of the microbiology of nitrogen cycle processes in aquaculture ecosystem, and will help optimize aquatic tailwater treatment strategies.
Keywords: NirS gene; Aquaculture ecosystem; Coastal high-altitude aquaculture pond; Denitrifying bacteria; Suspended particulate matter.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Particle associated denitrification in coastal aquaculture water.BMC Microbiol. 2025 Jul 2;25(1):396. doi: 10.1186/s12866-025-04122-0. BMC Microbiol. 2025. PMID: 40604432 Free PMC article.
-
[Denitrification Rates and nirS-type Denitrifying Bacteria Community Structure Characteristics of Bulk and Rhizosphere Soil in Spring and Summer in the Alpine Wetlands of the Qinghai-Tibet Plateau].Huan Jing Ke Xue. 2021 Oct 8;42(10):4959-4967. doi: 10.13227/j.hjkx.202101258. Huan Jing Ke Xue. 2021. PMID: 34581140 Chinese.
-
NirS-type denitrifying bacteria in aerobic water layers of two drinking water reservoirs: Insights into the abundance, community diversity and co-existence model.J Environ Sci (China). 2023 Feb;124:215-226. doi: 10.1016/j.jes.2021.10.013. Epub 2022 Feb 2. J Environ Sci (China). 2023. PMID: 36182133
-
[Characteristic Analysis of nirS Denitrifying Bacterial Community in Lijiahe Reservoir During Stratification].Huan Jing Ke Xue. 2022 Jan 8;43(1):306-313. doi: 10.13227/j.hjkx.202104037. Huan Jing Ke Xue. 2022. PMID: 34989515 Chinese.
-
A review on nirS-type and nirK-type denitrifiers via a scientometric approach coupled with case studies.Environ Sci Process Impacts. 2022 Feb 23;24(2):221-232. doi: 10.1039/d1em00518a. Environ Sci Process Impacts. 2022. PMID: 35072673 Review.
Cited by
-
Combined effects of acidification and warming on soil denitrification and microbial community.Front Microbiol. 2025 Apr 2;16:1572497. doi: 10.3389/fmicb.2025.1572497. eCollection 2025. Front Microbiol. 2025. PMID: 40241727 Free PMC article.
-
Particle associated denitrification in coastal aquaculture water.BMC Microbiol. 2025 Jul 2;25(1):396. doi: 10.1186/s12866-025-04122-0. BMC Microbiol. 2025. PMID: 40604432 Free PMC article.
References
-
- Choi M, et al. Strain and dose infectivity of Vibrio parahaemolyticus: The causative agent of early mortality syndrome in shrimp. Aquac. Res. 2017 doi: 10.1111/are.13197. - DOI
MeSH terms
Substances
Grants and funding
- NO. 42006137/National Natural Science Foundation of China
- NO. 42006137/National Natural Science Foundation of China
- NO. 42006137/National Natural Science Foundation of China
- NO. 42006137/National Natural Science Foundation of China
- NO. 42006137/National Natural Science Foundation of China
- No. 2020A1515110597/Basic and Applied Basic Research Foundation of Guangdong Province
- No. 2020A1515110597/Basic and Applied Basic Research Foundation of Guangdong Province
- No. 2020A1515110597/Basic and Applied Basic Research Foundation of Guangdong Province
- No. 2020A1515110597/Basic and Applied Basic Research Foundation of Guangdong Province
- No. 2020A1515110597/Basic and Applied Basic Research Foundation of Guangdong Province
LinkOut - more resources
Full Text Sources

