Sequential neoadjuvant chemotherapy using pegylated liposomal doxorubicin and cyclophosphamide followed by taxanes with complete trastuzumab and pertuzumab treatment for HER2-positive breast cancer: A phase II single-arm study
- PMID: 38455369
- PMCID: PMC10915636
- DOI: 10.21147/j.issn.1000-9604.2024.01.06
Sequential neoadjuvant chemotherapy using pegylated liposomal doxorubicin and cyclophosphamide followed by taxanes with complete trastuzumab and pertuzumab treatment for HER2-positive breast cancer: A phase II single-arm study
Abstract
Objective: Despite cardiotoxicity overlap, the trastuzumab/pertuzumab and anthracycline combination remains crucial due to significant benefits. Pegylated liposomal doxorubicin (PLD), a less cardiotoxic anthracycline, was evaluated for efficacy and cardiac safety when combined with cyclophosphamide and followed by taxanes with trastuzumab/pertuzumab in human epidermal growth factor receptor-2 (HER2)-positive early breast cancer (BC).
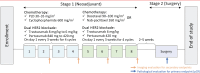
Methods: In this multicenter, phase II study, patients with confirmed HER2-positive early BC received four cycles of PLD (30-35 mg/m2) and cyclophosphamide (600 mg/m2), followed by four cycles of taxanes (docetaxel, 90-100 mg/m2 or nab-paclitaxel, 260 mg/m2), concomitant with eight cycles of trastuzumab (8 mg/kg loading dose, then 6 mg/kg) and pertuzumab (840 mg loading dose, then 420 mg) every 3 weeks. The primary endpoint was total pathological complete response (tpCR, ypT0/is ypN0). Secondary endpoints included breast pCR (bpCR), objective response rate (ORR), disease control rate, rate of breast-conserving surgery (BCS), and safety (with a focus on cardiotoxicity).
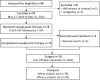
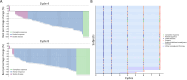
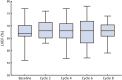
Results: Between May 27, 2020 and May 11, 2022, 78 patients were treated with surgery, 42 (53.8%) of whom had BCS. After neoadjuvant therapy, 47 [60.3%, 95% confidence interval (95% CI), 48.5%-71.2%] patients achieved tpCR, and 49 (62.8%) achieved bpCR. ORRs were 76.9% (95% CI, 66.0%-85.7%) and 93.6% (95% CI, 85.7%-97.9%) after 4-cycle and 8-cycle neoadjuvant therapy, respectively. Nine (11.5%) patients experienced asymptomatic left ventricular ejection fraction (LVEF) reductions of ≥10% from baseline, all with a minimum value of >55%. No treatment-related abnormal cardiac function changes were observed in mean N-terminal pro-BNP (NT-proBNP), troponin I, or high-sensitivity troponin.
Conclusions: This dual HER2-blockade with sequential polychemotherapy showed promising activity with rapid tumor regression in HER2-positive BC. Importantly, this regimen showed an acceptable safety profile, especially a low risk of cardiac events, suggesting it as an attractive treatment approach with a favorable risk-benefit balance.
Keywords: Breast cancer; HER2-positive breast cancer; dual HER2 blockade; neoadjuvant therapy; sequential therapy.
Copyright ©2024 Chinese Journal of Cancer Research. All rights reserved.
Figures
Similar articles
-
Pegylated Liposomal Doxorubicin, Docetaxel, and Trastuzumab as Neoadjuvant Treatment for HER2-Positive Breast Cancer Patients: A Phase II and Biomarker Study.Front Oncol. 2022 Jul 8;12:909426. doi: 10.3389/fonc.2022.909426. eCollection 2022. Front Oncol. 2022. PMID: 35875123 Free PMC article.
-
A Phase II Study of Neoadjuvant PLD/Cyclophosphamide and Sequential nab-Paclitaxel Plus Dual HER2 Blockade in HER2-Positive Breast Cancer.Oncologist. 2024 Jan 5;29(1):e15-e24. doi: 10.1093/oncolo/oyad160. Oncologist. 2024. PMID: 37279780 Free PMC article. Clinical Trial.
-
Pathologic Complete Response with Neoadjuvant Doxorubicin and Cyclophosphamide Followed by Paclitaxel with Trastuzumab and Pertuzumab in Patients with HER2-Positive Early Stage Breast Cancer: A Single Center Experience.Oncologist. 2017 Feb;22(2):139-143. doi: 10.1634/theoncologist.2016-0268. Epub 2017 Feb 6. Oncologist. 2017. PMID: 28167568 Free PMC article.
-
Risk-based decision-making in the treatment of HER2-positive early breast cancer: Recommendations based on the current state of knowledge.Cancer Treat Rev. 2021 Sep;99:102229. doi: 10.1016/j.ctrv.2021.102229. Epub 2021 May 20. Cancer Treat Rev. 2021. PMID: 34139476 Review.
-
Pathologic Complete Response Achieved in Early-Stage HER2-Positive Breast Cancer After Neoadjuvant Therapy With Trastuzumab and Chemotherapy vs. Trastuzumab, Chemotherapy, and Pertuzumab: A Systematic Review and Meta-Analysis of Clinical Trials.Cureus. 2023 May 31;15(5):e39780. doi: 10.7759/cureus.39780. eCollection 2023 May. Cureus. 2023. PMID: 37398703 Free PMC article. Review.
Cited by
-
Deep learning-based multi-task prediction of response to neoadjuvant chemotherapy using multiscale whole slide images in breast cancer: A multicenter study.Chin J Cancer Res. 2025 Jan 30;37(1):28-47. doi: 10.21147/j.issn.1000-9604.2025.01.03. Chin J Cancer Res. 2025. PMID: 40078559 Free PMC article.
References
-
- Xiang H, Xin L, Ye J, et al A multicenter study on efficacy of dual-target neoadjuvant therapy for HER2-positive breast cancer and a consistent analysis of efficacy evaluation of neoadjuvant therapy by Miller-Payne and RCB pathological evaluation systems (CSBrS-026) Chin J Cancer Res. 2023;35:702–12. doi: 10.21147/j.issn.1000-9604.2023.06.13. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
