Renal-Limited Thrombotic Microangiopathy in a Patient Who Received Gemcitabine, Ramucirumab, and Pembrolizumab: A Case Report and Literature Review
- PMID: 38455838
- PMCID: PMC10918209
- DOI: 10.7759/cureus.53669
Renal-Limited Thrombotic Microangiopathy in a Patient Who Received Gemcitabine, Ramucirumab, and Pembrolizumab: A Case Report and Literature Review
Abstract
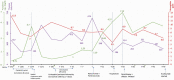
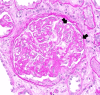
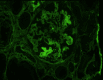
Cancer drug-induced thrombotic microangiopathy (DITMA) is an important and serious cause of kidney disease in cancer patients. In addition to classical chemotherapy, the increasing use of targeted therapy and immunotherapy has led to more oncotherapy-associated thrombotic microangiopathy (TMA). It is important for clinicians to recognize this potentially life-threatening adverse effect and gain knowledge of the patient's clinical course and treatment response. In this paper, we report a patient with lung cancer, who was treated with three different classes of anti-neoplastic agents, gemcitabine, ramucirumab, and pembrolizumab. This patient subsequently developed renal-limited thrombotic microangiopathy(rTMA) requiring hemodialysis. The varying features of TMA caused by these therapies were discussed. We also described the clinical course, diagnostic challenges, and management of this patient.
Keywords: acute kidney injury (aki); anti-neoplastic therapy; cancer drug induced tma; cancer treatment side effects; thrombotic microangiopathy (tma).
Copyright © 2024, Patel et al.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
Similar articles
-
Drug-induced thrombotic microangiopathy: An updated review of causative drugs, pathophysiology, and management.Front Pharmacol. 2023 Jan 9;13:1088031. doi: 10.3389/fphar.2022.1088031. eCollection 2022. Front Pharmacol. 2023. PMID: 36699080 Free PMC article. Review.
-
Gemcitabine-Induced Thrombotic Microangiopathy in a Patient With Cholangiocarcinoma: An Atypical Case.Cureus. 2024 Jun 28;16(6):e63385. doi: 10.7759/cureus.63385. eCollection 2024 Jun. Cureus. 2024. PMID: 39070506 Free PMC article.
-
Reversible renal-limited thrombotic microangiopathy due to gemcitabine-dexamethasone-cisplatin therapy: a case report.BMC Nephrol. 2021 May 12;22(1):175. doi: 10.1186/s12882-021-02386-y. BMC Nephrol. 2021. PMID: 33980166 Free PMC article.
-
Gemcitabine-associated thrombotic microangiopathy in a patient with lung cancer: A case report.Oncol Lett. 2017 Mar;13(3):1201-1203. doi: 10.3892/ol.2017.5576. Epub 2017 Jan 5. Oncol Lett. 2017. PMID: 28454234 Free PMC article.
-
Renal Thrombotic Microangiopathy: A Review.Am J Kidney Dis. 2023 May;81(5):591-605. doi: 10.1053/j.ajkd.2022.10.014. Epub 2022 Dec 10. Am J Kidney Dis. 2023. PMID: 36509342 Review.
References
-
- Pembrolizumab-associated thrombotic microangiopathy in a patient with urothelial cancer: a case report and literature review. Hayata M, Shimanuki M, Ko T, et al. Ren Replace Ther. 2020;6:29.
Publication types
LinkOut - more resources
Full Text Sources
