A mitochondrial NADPH-cholesterol axis regulates extracellular vesicle biogenesis to support hematopoietic stem cell fate
- PMID: 38458178
- PMCID: PMC10957094
- DOI: 10.1016/j.stem.2024.02.004
A mitochondrial NADPH-cholesterol axis regulates extracellular vesicle biogenesis to support hematopoietic stem cell fate
Abstract
Mitochondrial fatty acid oxidation (FAO) is essential for hematopoietic stem cell (HSC) self-renewal; however, the mechanism by which mitochondrial metabolism controls HSC fate remains unknown. Here, we show that within the hematopoietic lineage, HSCs have the largest mitochondrial NADPH pools, which are required for proper HSC cell fate and homeostasis. Bioinformatic analysis of the HSC transcriptome, biochemical assays, and genetic inactivation of FAO all indicate that FAO-generated NADPH fuels cholesterol synthesis in HSCs. Interference with FAO disturbs the segregation of mitochondrial NADPH toward corresponding daughter cells upon single HSC division. Importantly, we have found that the FAO-NADPH-cholesterol axis drives extracellular vesicle (EV) biogenesis and release in HSCs, while inhibition of EV signaling impairs HSC self-renewal. These data reveal the existence of a mitochondrial NADPH-cholesterol axis for EV biogenesis that is required for hematopoietic homeostasis and highlight the non-stochastic nature of HSC fate determination.
Keywords: HSC self-renewal; NADPH; cholesterol; exosomes; extracellular vesicles; fate determination; fatty acid oxidation; hematopoietic stem cell; metabolism; mitochondria.
Copyright © 2024 The Author(s). Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests T.F. is a founder and equity holder in Generian Pharmaceuticals and Coloma Therapeutics. D.T.S. is a director and equity holder of Agios Pharmaceuticals, Magenta Therapeutics, Editas Medicines, Clear Creek Bio, and LifeVaultBio; he is a founder of Fate Therapeutics and Magenta Therapeutics and a consultant to FOG Pharma, VcanBio, and Flagship Pioneering.
Figures
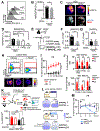



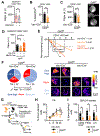
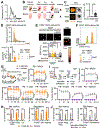
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

