Effective synthesis of high-content fructooligosaccharides in engineered Aspergillus niger
- PMID: 38461254
- PMCID: PMC10924377
- DOI: 10.1186/s12934-024-02353-w
Effective synthesis of high-content fructooligosaccharides in engineered Aspergillus niger
Abstract
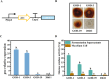
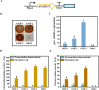
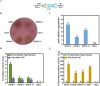
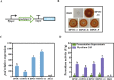
Background: Aspergillus niger ATCC 20611 is an industrially important fructooligosaccharides (FOS) producer since it produces the β-fructofuranosidase with superior transglycosylation activity, which is responsible for the conversion of sucrose to FOS accompanied by the by-product (glucose) generation. This study aims to consume glucose to enhance the content of FOS by heterologously expressing glucose oxidase and peroxidase in engineered A. niger.
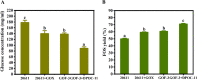
Results: Glucose oxidase was successfully expressed and co-localized with β-fructofuranosidase in mycelia. These mycelia were applied to synthesis of FOS, which possessed an increased purity of 60.63% from 52.07%. Furthermore, peroxidase was expressed in A. niger and reached 7.70 U/g, which could remove the potential inhibitor of glucose oxidase to facilitate the FOS synthesis. Finally, the glucose oxidase-expressing strain and the peroxidase-expressing strain were jointly used to synthesize FOS, which content achieved 71.00%.
Conclusions: This strategy allows for obtaining high-content FOS by the multiple enzymes expressed in the industrial fungus, avoiding additional purification processes used in the production of oligosaccharides. This study not only facilitated the high-purity FOS synthesis, but also demonstrated the potential of A. niger ATCC 20611 as an enzyme-producing cell factory.
Keywords: Aspergillus niger; Fructooligosaccharides; Glucose oxidase; Heterologous expression; Peroxidase; β-fructofuranosidase.
© 2024. The Author(s).
Conflict of interest statement
The authors declare that they have no competing of interests.
Figures
Similar articles
-
Establishment of a rapid and effective plate chromogenic assay for screening of Aspergillus species with high β-fructofuranosidase activity for fructooligosaccharides production.J Microbiol Methods. 2019 Nov;166:105740. doi: 10.1016/j.mimet.2019.105740. Epub 2019 Oct 12. J Microbiol Methods. 2019. PMID: 31614171
-
Continuous production of fructooligosaccharides by recycling of the thermal-stable β-fructofuranosidase produced by Aspergillus niger.Biotechnol Lett. 2021 Jun;43(6):1175-1182. doi: 10.1007/s10529-021-03099-w. Epub 2021 Feb 11. Biotechnol Lett. 2021. PMID: 33575897
-
High level production of a β-fructofuranosidase in Aspergillusniger for the preperation of prebiotic bread using in situ enzymatic conversion.Food Res Int. 2025 May;208:116225. doi: 10.1016/j.foodres.2025.116225. Epub 2025 Mar 15. Food Res Int. 2025. PMID: 40263796
-
Expression of an Aspergillus niger glucose oxidase in saccharomyces cerevisiae and its use to optimize fructo-oligosaccharides synthesis.Biotechnol Prog. 2006 Jul-Aug;22(4):1096-101. doi: 10.1021/bp060076k. Biotechnol Prog. 2006. PMID: 16889385
-
Gene expression in filamentous fungi. Expression of pectinases and glucose oxidase in Aspergillus niger.Bioprocess Technol. 1995;22:241-308. Bioprocess Technol. 1995. PMID: 7765534 Review. No abstract available.
Cited by
-
Optimization of the fermentation process for fructosyltransferase production by Aspergillus niger FS054.Microb Cell Fact. 2025 Jul 27;24(1):173. doi: 10.1186/s12934-025-02798-7. Microb Cell Fact. 2025. PMID: 40717065 Free PMC article.
-
Development of an efficient heterologous protein expression platform in Aspergillus niger through genetic modification of a glucoamylase hyperproducing industrial strain.Microb Cell Fact. 2025 Jul 8;24(1):160. doi: 10.1186/s12934-025-02786-x. Microb Cell Fact. 2025. PMID: 40629383 Free PMC article.
References
-
- Zeng J, Hu Y, Gao H, Sun J, Ma H. Fructo-oligosaccharides impact on the hydration and retro-gradation of wheat starch and gel. Int J Food Prop. 2016;19(12):2682–2692. doi: 10.1080/10942912.2015.1136937. - DOI
-
- Sangeetha PT, Ramesh MN, Prapulla SG. Recent trends in the production, analysis and application of fructooligosaccharides. Trends Food Sci Technol. 2005;16:442–457. doi: 10.1016/j.tifs.2005.05.003. - DOI
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources

