This is a preprint.
Zebrafish and cellular models of SELENON-Related Myopathy exhibit novel embryonic and metabolic phenotypes
- PMID: 38464009
- PMCID: PMC10925121
- DOI: 10.1101/2024.02.26.581979
Zebrafish and cellular models of SELENON-Related Myopathy exhibit novel embryonic and metabolic phenotypes
Update in
-
Zebrafish and cellular models of SELENON-Congenital myopathy exhibit novel embryonic and metabolic phenotypes.Skelet Muscle. 2025 Mar 15;15(1):7. doi: 10.1186/s13395-025-00376-4. Skelet Muscle. 2025. PMID: 40087793 Free PMC article.
Abstract
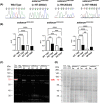
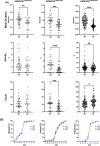
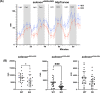
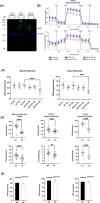
SELENON-Related Myopathy (SELENON-RM) is a rare congenital myopathy caused by mutations of the SELENON gene characterized by axial muscle weakness and progressive respiratory insufficiency. Muscle histopathology commonly includes multiminicores or a dystrophic pattern but is often non-specific. The SELENON gene encodes selenoprotein N (SelN), a selenocysteine-containing redox enzyme located in the endo/sarcoplasmic reticulum membrane where it colocalizes with mitochondria-associated membranes. However, the molecular mechanism(s) by which SelN deficiency causes SELENON-RM are undetermined. A hurdle is the lack of cellular and animal models that show assayable phenotypes. Here we report deep-phenotyping of SelN-deficient zebrafish and muscle cells. SelN-deficient zebrafish exhibit changes in embryonic muscle function and swimming activity in larvae. Analysis of single cell RNAseq data in a zebrafish embryo-atlas revealed coexpression between selenon and genes involved in glutathione redox pathway. SelN-deficient zebrafish and mouse myoblasts exhibit changes in glutathione and redox homeostasis, suggesting a direct relationship with SelN function. We report changes in metabolic function abnormalities in SelN-null myotubes when compared to WT. These results suggest that SelN has functional roles during zebrafish early development and myoblast metabolism.
Keywords: Selenoprotein N; congenital myopathy; multiminicore myopathy; rigid spine muscular dystrophy; zebrafish model.
Conflict of interest statement
Competing Interests AHB receives consulting income from Kate Therapeutics, Roche Pharmaceuticals, GLG Inc, and Guidepoint Global, and has equity in Kate Therapeutics and Kinea Bio. For all other authors no competing interests are declared.
Figures



References
-
- Arbogast S., Beuvin M., Fraysse B., Zhou H., Muntoni F. and Ferreiro A. (2009). Oxidative stress in SEPN1-related myopathy: From pathophysiology to treatment. Ann. Neurol. 65, 677–686. - PubMed
-
- Bouman K., Groothuis J. T., Doorduin J., van Alfen N., Udink ten Cate F. E. A., van den Heuvel F. M. A., Nijveldt R., van Tilburg W. C. M., Buckens S. C. F. M., Dittrich A. T. M., et al. (2021). Natural history, outcome measures and trial readiness in LAMA2-related muscular dystrophy and SELENON-related myopathy in children and adults: protocol of the LAST STRONG study. BMC Neurol. 21, 313. - PMC - PubMed
-
- Buss R. R. and Drapeau P. (2001). Synaptic drive to motoneurons during fictive swimming in the developing zebrafish. J. Neurophysiol. 86, 197–210. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
