This is a preprint.
Tunable metastability of condensates reconciles their dual roles in amyloid fibril formation
- PMID: 38464104
- PMCID: PMC10925303
- DOI: 10.1101/2024.02.28.582569
Tunable metastability of condensates reconciles their dual roles in amyloid fibril formation
Update in
-
Tunable metastability of condensates reconciles their dual roles in amyloid fibril formation.Mol Cell. 2025 Jun 5;85(11):2230-2245.e7. doi: 10.1016/j.molcel.2025.05.011. Epub 2025 May 28. Mol Cell. 2025. PMID: 40441157
Abstract
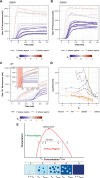
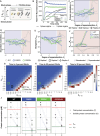
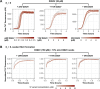
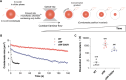
Stress granules form via co-condensation of RNA-binding proteins containing prion-like low complexity domains (PLCDs) with RNA molecules. Homotypic interactions among PLCDs can drive amyloid fibril formation that is enhanced by ALS-associated mutations. We report that condensation- versus fibril-driving homotypic interactions are separable for A1-LCD, the PLCD of hnRNPA1. Separable interactions lead to thermodynamically metastable condensates and globally stable fibrils. Interiors of condensates suppress fibril formation whereas interfaces have the opposite effect. ALS-associated mutations enhance the stability of fibrils and weaken condensate metastability, thus enhancing the rate of fibril formation. We designed mutations to enhance A1-LCD condensate metastability and discovered that stress granule disassembly in cells can be restored even when the designed variants carry ALS-causing mutations. Therefore, fibril formation can be suppressed by condensate interiors that function as sinks. Condensate sink potentials are influenced by their metastability, which is tunable through separable interactions even among minority components of stress granules.
Keywords: Phase separation; RNP granule; amyotrophic lateral sclerosis; fibril formation; frontotemporal dementia; prion-like domain; stress granule; supersaturation.
Conflict of interest statement
DECLARATION OF INTERESTS RVP is a member of the Scientific Advisory Board and a shareholder of Dewpoint Therapeutics. TM is a member of the advisory board of Molecular Cell. PRB is a member of the Biophysics Reviews (AIP Publishing) editorial board. The work reported here was not influenced by these affiliations. The remaining authors declare no competing interests.
Figures


Similar articles
-
Tunable metastability of condensates reconciles their dual roles in amyloid fibril formation.Mol Cell. 2025 Jun 5;85(11):2230-2245.e7. doi: 10.1016/j.molcel.2025.05.011. Epub 2025 May 28. Mol Cell. 2025. PMID: 40441157
-
Phase Separation in Mixtures of Prion-Like Low Complexity Domains is Driven by the Interplay of Homotypic and Heterotypic Interactions.bioRxiv [Preprint]. 2023 Mar 16:2023.03.15.532828. doi: 10.1101/2023.03.15.532828. bioRxiv. 2023. Update in: Nat Commun. 2023 Sep 8;14(1):5527. doi: 10.1038/s41467-023-41274-x. PMID: 36993212 Free PMC article. Updated. Preprint.
-
Phase Separation in Mixtures of Prion-Like Low Complexity Domains is Driven by the Interplay of Homotypic and Heterotypic Interactions.Res Sq [Preprint]. 2023 May 4:rs.3.rs-2870258. doi: 10.21203/rs.3.rs-2870258/v1. Res Sq. 2023. Update in: Nat Commun. 2023 Sep 8;14(1):5527. doi: 10.1038/s41467-023-41274-x. PMID: 37205474 Free PMC article. Updated. Preprint.
-
Phase separation and pathologic transitions of RNP condensates in neurons: implications for amyotrophic lateral sclerosis, frontotemporal dementia and other neurodegenerative disorders.Front Mol Neurosci. 2023 Sep 1;16:1242925. doi: 10.3389/fnmol.2023.1242925. eCollection 2023. Front Mol Neurosci. 2023. PMID: 37720552 Free PMC article. Review.
-
From Prions to Stress Granules: Defining the Compositional Features of Prion-Like Domains That Promote Different Types of Assemblies.Int J Mol Sci. 2021 Jan 27;22(3):1251. doi: 10.3390/ijms22031251. Int J Mol Sci. 2021. PMID: 33513942 Free PMC article. Review.
References
-
- Guillen-Boixet J., Kopach A., Holehouse A.S., Wittmann S., Jahnel M., Schlussler R., Kim K., Trussina I., Wang J., Mateju D., et al. (2020). RNA-Induced Conformational Switching and Clustering of G3BP Drive Stress Granule Assembly by Condensation. Cell 181, 346–361 e317. 10.1016/j.cell.2020.03.049. - DOI - PMC - PubMed
-
- Van Treeck B., Protter D.S.W., Matheny T., Khong A., Link C.D., and Parker R. (2018). RNA self-assembly contributes to stress granule formation and defining the stress granule transcriptome. Proceedings of the National Academy of Sciences 115, 2734–2739. 10.1073/pnas.1800038115. - DOI - PMC - PubMed
-
- Wang J., Choi J.-M., Holehouse A.S., Lee H.O., Zhang X., Jahnel M., Maharana S., Lemaitre R., Pozniakovsky A., Drechsel D., et al. (2018). A Molecular Grammar Governing the Driving Forces for Phase Separation of Prion-like RNA Binding Proteins. Cell 174, 688–699.e616. 10.1016/j.cell.2018.06.006. - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
