Leaf variegation caused by plastome structural variation: an example from Dianella tasmanica
- PMID: 38464478
- PMCID: PMC10923649
- DOI: 10.1093/hr/uhae009
Leaf variegation caused by plastome structural variation: an example from Dianella tasmanica
Abstract
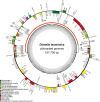
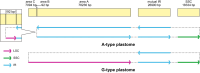
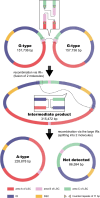
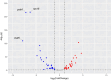
Variegated plants often exhibit plastomic heteroplasmy due to single-nucleotide mutations or small insertions/deletions in their albino sectors. Here, however, we identified a plastome structural variation in albino sectors of the variegated plant Dianella tasmanica (Asphodelaceae), a perennial herbaceous plant widely cultivated as an ornamental in tropical Asia. This structural variation, caused by intermolecular recombination mediated by an 11-bp inverted repeat flanking a 92-bp segment in the large single-copy region (LSC), generates a giant plastome (228 878 bp) with the largest inverted repeat of 105 226 bp and the smallest LSC of 92 bp known in land plants. It also generates an ~7-kb deletion on the boundary of the LSC, which eliminates three protein coding genes (psbA, matK, and rps16) and one tRNA gene (trnK). Albino sectors exhibit dramatic changes in expression of many plastid genes, including negligible expression of psbA, matK, and rps16, reduced expression of photosynthesis-related genes, and increased expression of genes related to the translational apparatus. Microscopic and ultrastructure observations showed that albino tissues were present in both green and albino sectors of the variegated individuals, and chloroplasts were poorly developed in the mesophyll cells of the albino tissues of the variegated individuals. These poorly developed chloroplasts likely carry the large and rearranged plastome, which is likely responsible for the loss of photosynthesis and albinism in the leaf margins. Considering that short repeats are relatively common in plant plastomes and that photosynthesis is not necessary for albino sectors, structural variation of this kind may not be rare in the plastomes of variegated plants.
© The Author(s) 2024. Published by Oxford University Press on behalf of Nanjing Agricultural University.
Conflict of interest statement
No conflict of interest declared.
Figures


Similar articles
-
Evolutionary differences in gene loss and pseudogenization among mycoheterotrophic orchids in the tribe Vanilleae (subfamily Vanilloideae).Front Plant Sci. 2023 Mar 22;14:1160446. doi: 10.3389/fpls.2023.1160446. eCollection 2023. Front Plant Sci. 2023. PMID: 37035052 Free PMC article.
-
Comprehensive genomic analyses with 115 plastomes from algae to seed plants: structure, gene contents, GC contents, and introns.Genes Genomics. 2020 May;42(5):553-570. doi: 10.1007/s13258-020-00923-x. Epub 2020 Mar 21. Genes Genomics. 2020. PMID: 32200544
-
Fine Mapping of BoVl Conferring the Variegated Leaf in Ornamental Kale (Brassica oleracea var. acephala).Int J Mol Sci. 2022 Nov 27;23(23):14853. doi: 10.3390/ijms232314853. Int J Mol Sci. 2022. PMID: 36499179 Free PMC article.
-
High-throughput discovery of plastid genes causing albino phenotypes in ornamental chimeric plants.Hortic Res. 2022 Nov 3;10(1):uhac246. doi: 10.1093/hr/uhac246. eCollection 2023. Hortic Res. 2022. PMID: 36643742 Free PMC article.
-
Leaf-variegated mutations and their responsible genes in Arabidopsis thaliana.Genes Genet Syst. 2003 Feb;78(1):1-9. doi: 10.1266/ggs.78.1. Genes Genet Syst. 2003. PMID: 12655133 Review.
Cited by
-
Integrated genomic and transcriptomic analysis reveals the mechanisms underlying leaf variegation in 'Gonggan' mandarin.BMC Plant Biol. 2025 Apr 15;25(1):472. doi: 10.1186/s12870-025-06496-9. BMC Plant Biol. 2025. PMID: 40229686 Free PMC article.
-
The CsPPR gene with RNA-editing function involved in leaf color asymmetry of the reciprocal hybrids derived from Cucumis sativus and C. hystrix.Planta. 2024 Sep 20;260(4):102. doi: 10.1007/s00425-024-04513-z. Planta. 2024. PMID: 39302471
-
A recent large-scale intraspecific IR expansion and evolutionary dynamics of the plastome of Peucedanum japonicum.Sci Rep. 2025 Jan 2;15(1):104. doi: 10.1038/s41598-024-84540-8. Sci Rep. 2025. PMID: 39748098 Free PMC article.
-
Transcriptomic and Structural Insights into Leaf Variegation Development in Ilex × 'Solar Flare'.Int J Mol Sci. 2025 Apr 23;26(9):3999. doi: 10.3390/ijms26093999. Int J Mol Sci. 2025. PMID: 40362242 Free PMC article.
References
-
- Adir N, Zer H, Shochat S. et al. . Photoinhibition – a historical perspective. Photosynth Res. 2003;76:343–70 - PubMed
-
- Azarin K, Usatov A, Makarenko M. et al. . A point mutation in the photosystem I P700 chlorophyll a apoprotein A1 gene confers variegation in Helianthus annuus L. Plant Mol Biol. 2020;103:373–89 - PubMed
-
- Bain JF, Jansen RK. A chloroplast DNA hairpin structure provides useful phylogenetic data within tribe Senecioneae (Asteraceae). Can J Bot. 2006;84:862–8
LinkOut - more resources
Full Text Sources
Miscellaneous

