Growth capacity of a Wharton's Jelly derived mesenchymal stromal cells tissue engineered vascular graft used for main pulmonary artery reconstruction in piglets
- PMID: 38464540
- PMCID: PMC10920298
- DOI: 10.3389/fbioe.2024.1360221
Growth capacity of a Wharton's Jelly derived mesenchymal stromal cells tissue engineered vascular graft used for main pulmonary artery reconstruction in piglets
Abstract
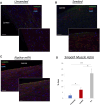
Background: Surgical treatment of congenital heart defects affecting the right ventricular outflow tract (RVOT) often requires complex reconstruction and multiple reoperations due to structural degeneration and lack of growth of currently available materials. Hence, alternative approaches for RVOT reconstruction, which meet the requirements of biocompatibility and long-term durability of an ideal scaffold, are needed. Through this full scale pre-clinical study, we demonstrated the growth capacity of a Wharton's Jelly derived mesenchymal stromal cells (WJ-MSC) tissue engineered vascular graft used in reconstructing the main pulmonary artery in piglets, providing proof of biocompatibility and efficacy. Methods: Sixteen four-week-old Landrace pigs were randomized to undergo supravalvar Main Pulmonary Artery (MPA) replacement with either unseeded or WJ-MSCs-seeded Small Intestinal Submucosa-derived grafts. Animals were followed up for 6 months by clinical examinations and cardiac imaging. At termination, sections of MPAs were assessed by macroscopic inspection, histology and fluorescent immunohistochemistry. Results: Data collected at 6 months follow up showed no sign of graft thrombosis or calcification. The explanted main pulmonary arteries demonstrated a significantly higher degree of cellular organization and elastin content in the WJ-MSCs seeded grafts compared to the acellular counterparts. Transthoracic echocardiography and cardiovascular magnetic resonance confirmed the superior growth and remodelling of the WJ-MSCs seeded conduit compared to the unseeded. Conclusion: Our findings indicate that the addition of WJ-MSCs to the acellular scaffold can upgrade the material, converting it into a biologically active tissue, with the potential to grow, repair and remodel the RVOT.
Keywords: growing swine model; preclinical efficacy; right ventricular outflow tract reconstruction; small intestinal submucosa; tissue engineering.
Copyright © 2024 Iacobazzi, Ghorbel, Rapetto, Narayan, Deutsch, Salih, Harris, Skeffington, Parry, Parolari, Chanoit and Caputo.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures




References
-
- Albertario A., Swim M. M., Ahmed E. M., Iacobazzi D., Yeong M., Madeddu P., et al. (2019). Successful reconstruction of the right ventricular outflow tract by implantation of thymus stem cell engineered graft in growing swine. JACC Basic Transl. Sci. 4 (3), 364–384. 10.1016/j.jacbts.2019.02.001 - DOI - PMC - PubMed
-
- Brown J. W., Ruzmetov M., Rodefeld M. D., Vijay P., Turrentine M. W. (2005). Right ventricular outflow tract reconstruction with an allograft conduit in non-ross patients: risk factors for allograft dysfunction and failure. Ann. Thorac. Surg. 80 (2), 655–664. 10.1016/j.athoracsur.2005.02.053 - DOI - PubMed
-
- Cunnane E. M., Lorentz K. L., Soletti L., Ramaswamy A. K., Chung T. K., Haskett D. G., et al. (2020b). Development of a semi-automated, bulk seeding Device for large animal model implantation of tissue engineered vascular grafts. Front. Bioeng. Biotechnol. 8, 597847. 10.3389/fbioe.2020.597847 - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

