Acute Aromatase Inhibition Impairs Neural and Behavioral Auditory Scene Analysis in Zebra Finches
- PMID: 38467426
- PMCID: PMC10960633
- DOI: 10.1523/ENEURO.0423-23.2024
Acute Aromatase Inhibition Impairs Neural and Behavioral Auditory Scene Analysis in Zebra Finches
Abstract
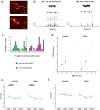
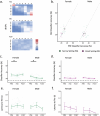
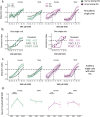
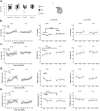
Auditory perception can be significantly disrupted by noise. To discriminate sounds from noise, auditory scene analysis (ASA) extracts the functionally relevant sounds from acoustic input. The zebra finch communicates in noisy environments. Neurons in their secondary auditory pallial cortex (caudomedial nidopallium, NCM) can encode song from background chorus, or scenes, and this capacity may aid behavioral ASA. Furthermore, song processing is modulated by the rapid synthesis of neuroestrogens when hearing conspecific song. To examine whether neuroestrogens support neural and behavioral ASA in both sexes, we retrodialyzed fadrozole (aromatase inhibitor, FAD) and recorded in vivo awake extracellular NCM responses to songs and scenes. We found that FAD affected neural encoding of songs by decreasing responsiveness and timing reliability in inhibitory (narrow-spiking), but not in excitatory (broad-spiking) neurons. Congruently, FAD decreased neural encoding of songs in scenes for both cell types, particularly in females. Behaviorally, we trained birds using operant conditioning and tested their ability to detect songs in scenes after administering FAD orally or injected bilaterally into NCM. Oral FAD increased response bias and decreased correct rejections in females, but not in males. FAD in NCM did not affect performance. Thus, FAD in the NCM impaired neuronal ASA but that did not lead to behavioral disruption suggesting the existence of resilience or compensatory responses. Moreover, impaired performance after systemic FAD suggests involvement of other aromatase-rich networks outside the auditory pathway in ASA. This work highlights how transient estrogen synthesis disruption can modulate higher-order processing in an animal model of vocal communication.
Keywords: aromatase inhibition; auditory scene analysis; caudomedial nidopallium; extracellular recordings; operant conditioning; zebra finches.
Copyright © 2024 Fernández-Vargas et al.
Conflict of interest statement
The authors declare no competing financial interests.
Figures



References
-
- Anand K, Nealen PM (2019) Differential GO and NOGO learning within an auditory discrimination task. Anim Behav Cogn 6:141–157. 10.26451/abc.06.02.05.2019 - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
