The transcriptomic insight into the differential susceptibility of African Swine Fever in inbred pigs
- PMID: 38467747
- PMCID: PMC10928096
- DOI: 10.1038/s41598-024-56569-2
The transcriptomic insight into the differential susceptibility of African Swine Fever in inbred pigs
Abstract
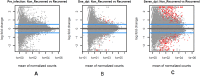
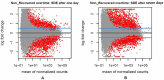
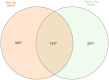
African swine fever (ASF) is a global threat to animal health and food security. ASF is typically controlled by strict biosecurity, rapid diagnosis, and culling of affected herds. Much progress has been made in developing modified live virus vaccines against ASF. There is host variation in response to ASF infection in the field and under controlled conditions. To better understand the dynamics underlying this host differential morbidity, whole transcriptome profiling was carried out in twelve immunized and five sham immunized pigs. Seventeen MHC homozygous inbred Large white Babraham pigs were sampled at three time points before and after the challenge. The changes in the transcriptome profiles of infected animals were surveyed over time. In addition, the immunization effect on the host response was studied as well among the contrasts of all protection subgroups. The results showed two promising candidate genes to distinguish between recovered and non-recovered pigs after infection with a virulent African swine fever virus (ASFV) pre-infection: HTRA3 and GFPT2 (padj < 0.05). Variant calling on the transcriptome assemblies showed a two-base pair insertion into the ACOX3 gene closely located to HTRA3 that may regulate its expression as a putative genomic variant for ASF. Several significant DGEs, enriched gene ontology (GO) terms, and KEGG pathways at 1 day and 7 days post-infection, compared to the pre-infection, indicate a significant inflammation response immediately after ASF infection. The presence of the virus was confirmed by the mapping of RNA-Seq reads on two whole viral genome sequences. This was concordant with a higher virus load in the non-recovered animals 7 days post-infection. There was no transcriptome signature on the immunization at pre-infection and 1 day post-infection. More samples and data from additional clinical trials may support these findings.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures




Similar articles
-
A QP509L/QP383R-Deleted African Swine Fever Virus Is Highly Attenuated in Swine but Does Not Confer Protection against Parental Virus Challenge.J Virol. 2022 Jan 12;96(1):e0150021. doi: 10.1128/JVI.01500-21. Epub 2021 Oct 6. J Virol. 2022. PMID: 34613824 Free PMC article.
-
Transcriptome profile of spleen tissues from locally-adapted Kenyan pigs (Sus scrofa) experimentally infected with three varying doses of a highly virulent African swine fever virus genotype IX isolate: Ken12/busia.1 (ken-1033).BMC Genomics. 2022 Jul 19;23(1):522. doi: 10.1186/s12864-022-08754-8. BMC Genomics. 2022. PMID: 35854219 Free PMC article.
-
Small RNA sequencing and profiling of serum-derived exosomes from African swine fever virus-infected pigs.J Anim Sci. 2023 Jan 3;101:skac400. doi: 10.1093/jas/skac400. J Anim Sci. 2023. PMID: 36478238 Free PMC article.
-
Transcriptome signatures of host tissue infected with African swine fever virus reveal differential expression of associated oncogenes.Arch Virol. 2024 Feb 21;169(3):54. doi: 10.1007/s00705-023-05959-4. Arch Virol. 2024. PMID: 38381218 Review.
-
[African swine fever in Russian Federation].Vopr Virusol. 2012 Sep-Oct;57(5):4-10. Vopr Virusol. 2012. PMID: 23248852 Review. Russian.
Cited by
-
Monoclonal Antibodies Targeting Porcine Macrophages Are Able to Inhibit the Cell Entry of Macrophage-Tropic Viruses (PRRSV and ASFV).Viruses. 2025 Jan 24;17(2):167. doi: 10.3390/v17020167. Viruses. 2025. PMID: 40006922 Free PMC article.
-
Advances in bioinformatics and multi-omics integration: transforming viral infectious disease research in veterinary medicine.Virol J. 2025 Jan 31;22(1):22. doi: 10.1186/s12985-025-02640-x. Virol J. 2025. PMID: 39891257 Free PMC article. Review.
References
-
- (WOAH), W. O. f. A. H. 35th Situation Report on African Swine Fever (ASF). (2023).
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

