The methodological quality of systematic reviews regarding the Core Outcome Set (COS) development
- PMID: 38468223
- PMCID: PMC10926669
- DOI: 10.1186/s12874-024-02182-w
The methodological quality of systematic reviews regarding the Core Outcome Set (COS) development
Abstract
Background: The Core Outcome Measures in Effectiveness Trials (COMET) working group proposed core outcome sets (COS) to address the heterogeneity in outcome measures in clinical studies. According to the recommendations of COMET, performing systematic reviews (SRs) usually was the first step for COS development. However, the SRs that serve as a basis for COS are not specifically appraised by organizations such as COMET regarding their quality. Here, we investigated the status of SRs related to development of COS and evaluated their methodological quality.
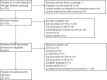
Methods: We conducted a search on PubMed to identify SRs related to COS development published from inception to May 2022. We qualitatively summarized the disease included in SR topics, and the studies included in the SRs. We evaluated the methodological quality of the SRs using AMSTAR 2.0 and compared the overall quality of SRs with and without protocols using the Mann-Whitney U test.
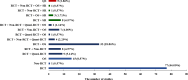
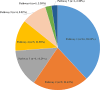
Results: We included 175 SRs from 23 different countries or regions, and they mainly focused on five diseases: musculoskeletal system or connective tissue disease (n = 19, 10.86%), injury, poisoning, or certain other consequences of external causes (n = 18, 10.29%), digestive system disease (n = 16, 9.14%), nervous system disease (n = 15, 8.57%), and genitourinary system disease (n = 15, 8.57%). Although 88.00% of SRs included randomized controlled trials (RCTs), only a few SRs (23.38%) employed appropriate tools to assess the risk of bias in RCTs. The assessment results on the basis of AMSTAR 2.0 indicated that most SRs (93.71%) were rated as ''critically low'' to ''low'' in terms of overall confidence. The overall confidence of SRs with protocols was significantly higher than that without protocols (P <.001). Compared to the SRs with protocols on Core Outcome Measures in Effectiveness Trials (COMET), SRs with protocols on PROSPERO were of better overall confidence (P = .017).
Conclusion: The overall quality of published SRs regarding COS development was poor. Our findings emphasize the need for researchers to carefully select the disease topic and strictly adhere to the requirements of optimal methodology when conducting a SR for the establishment of a COS.
Keywords: AMSATR 2.0; Core outcome measures in effectiveness trials (COMET); Core outcome set (COS); Methodological research; Systematic reviews (SRs).
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
The future of Cochrane Neonatal.Early Hum Dev. 2020 Nov;150:105191. doi: 10.1016/j.earlhumdev.2020.105191. Epub 2020 Sep 12. Early Hum Dev. 2020. PMID: 33036834
-
Study found increasing use of core outcome sets in Cochrane systematic reviews and identified facilitators and barriers.J Clin Epidemiol. 2024 May;169:111277. doi: 10.1016/j.jclinepi.2024.111277. Epub 2024 Feb 29. J Clin Epidemiol. 2024. PMID: 38428540
-
Core outcomes were rarely reported overall in systematic reviews on acupuncture for osteoarthritis: a cross-sectional meta-epidemiological study.Acupunct Med. 2023 Jun;41(3):142-150. doi: 10.1177/09645284221108215. Epub 2022 Aug 19. Acupunct Med. 2023. PMID: 35983785
-
Effectiveness and safety of manual therapy for knee osteoarthritis: An overview of systematic reviews and meta-analyses.Front Public Health. 2023 Feb 24;11:1081238. doi: 10.3389/fpubh.2023.1081238. eCollection 2023. Front Public Health. 2023. PMID: 36908468 Free PMC article.
-
More than half of systematic reviews have relevant core outcome sets.J Clin Epidemiol. 2021 Aug;136:168-179. doi: 10.1016/j.jclinepi.2021.04.019. Epub 2021 May 8. J Clin Epidemiol. 2021. PMID: 33974970 Free PMC article. Review.
References
MeSH terms
Grants and funding
- No.72064004/National Natural Science Foundation of China
- GZSYBS (2019) No.09/Doctoral Foundation of Guizhou Provincial People's Hospital
- gzwkj2022-224/Foundation of Health Commission of Guizhou Province
- CPA-Z05-ZC-2023-002/The Hospital Pharmaceutical Research Project of China Pharmaceutical Association
LinkOut - more resources
Full Text Sources
Research Materials

