The interactions between ineffective erythropoiesis and ferroptosis in β-thalassemia
- PMID: 38468700
- PMCID: PMC10925657
- DOI: 10.3389/fphys.2024.1346173
The interactions between ineffective erythropoiesis and ferroptosis in β-thalassemia
Abstract
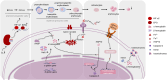
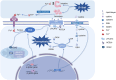
In Guangxi, Hainan, and Fujian Province in southern China, β-thalassemia is a frequent monogenic hereditary disorder that is primarily defined by hemolytic anemia brought on by inefficient erythropoiesis. It has been found that ineffective erythropoiesis in β-thalassemia is closely associated with a high accumulation of Reactive oxygen species, a product of oxidative stress, in erythroid cells. During recent years, ferroptosis is an iron-dependent lipid peroxidation that involves abnormalities in lipid and iron metabolism as well as reactive oxygen species homeostasis. It is a recently identified kind of programmed cell death. β-thalassemia patients experience increased iron release from reticuloendothelial cells and intestinal absorption of iron, ultimately resulting in iron overload. Additionally, the secretion of Hepcidin is inhibited in these patients. What counts is both ineffective erythropoiesis and ferroptosis in β-thalassemia are intricately linked to the iron metabolism and Reactive oxygen species homeostasis. Consequently, to shed further light on the pathophysiology of β-thalassemia and propose fresh ideas for its therapy, this paper reviews ferroptosis, ineffective erythropoiesis, and the way they interact.
Keywords: ROS; ferroptosis; ineffective erythropoiesis; iron overload; pathogenesis; β-thalassemia.
Copyright © 2024 Lin, Zheng, Chen, Xu and Huang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Oral ferroportin inhibitor vamifeport for improving iron homeostasis and erythropoiesis in β-thalassemia: current evidence and future clinical development.Expert Rev Hematol. 2021 Jul;14(7):633-644. doi: 10.1080/17474086.2021.1935854. Epub 2021 Jul 29. Expert Rev Hematol. 2021. PMID: 34324404 Review.
-
Ferroportin inhibitor vamifeport ameliorates ineffective erythropoiesis in a mouse model of β-thalassemia with blood transfusions.Haematologica. 2023 Oct 1;108(10):2703-2714. doi: 10.3324/haematol.2022.282328. Haematologica. 2023. PMID: 37165842 Free PMC article.
-
β-thalassemia: a model for elucidating the dynamic regulation of ineffective erythropoiesis and iron metabolism.Blood. 2011 Oct 20;118(16):4321-30. doi: 10.1182/blood-2011-03-283614. Epub 2011 Jul 18. Blood. 2011. PMID: 21768301 Free PMC article. Review.
-
Hepcidin and Anemia: A Tight Relationship.Front Physiol. 2019 Oct 9;10:1294. doi: 10.3389/fphys.2019.01294. eCollection 2019. Front Physiol. 2019. PMID: 31649559 Free PMC article. Review.
-
Imbalance of erythropoiesis and iron metabolism in patients with thalassemia.Int J Med Sci. 2019 Jan 1;16(2):302-310. doi: 10.7150/ijms.27829. eCollection 2019. Int J Med Sci. 2019. PMID: 30745811 Free PMC article.
Cited by
-
Ferroptosis as an emerging target in sickle cell disease.Curr Res Toxicol. 2024 Jun 18;7:100181. doi: 10.1016/j.crtox.2024.100181. eCollection 2024. Curr Res Toxicol. 2024. PMID: 39021403 Free PMC article.
-
Association Between Ferroptosis and Acute Kidney Disease (AKD): Unveiling the Expression of Genes Related to Iron Regulatory Metabolism.Biochem Genet. 2025 Jul 18. doi: 10.1007/s10528-025-11188-y. Online ahead of print. Biochem Genet. 2025. PMID: 40679656
-
Cell death signaling in human erythron: erythrocytes lose the complexity of cell death machinery upon maturation.Apoptosis. 2025 Apr;30(3-4):652-673. doi: 10.1007/s10495-025-02081-5. Epub 2025 Feb 9. Apoptosis. 2025. PMID: 39924584 Free PMC article. Review.
-
The 1357 bp deletion in β-thalassemia: molecular profiling and hematological characterization in a Guangxi cohort.Mol Biol Rep. 2025 Jun 16;52(1):602. doi: 10.1007/s11033-025-10724-8. Mol Biol Rep. 2025. PMID: 40522534
-
Iron-Loading Anemias.Adv Exp Med Biol. 2025;1480:145-161. doi: 10.1007/978-3-031-92033-2_11. Adv Exp Med Biol. 2025. PMID: 40603790 Review.
References
Publication types
LinkOut - more resources
Full Text Sources

