Conservation Mitonuclear Replacement: Facilitated mitochondrial adaptation for a changing world
- PMID: 38468713
- PMCID: PMC10925831
- DOI: 10.1111/eva.13642
Conservation Mitonuclear Replacement: Facilitated mitochondrial adaptation for a changing world
Abstract
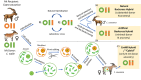
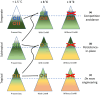
Most species will not be able to migrate fast enough to cope with climate change, nor evolve quickly enough with current levels of genetic variation. Exacerbating the problem are anthropogenic influences on adaptive potential, including the prevention of gene flow through habitat fragmentation and the erosion of genetic diversity in small, bottlenecked populations. Facilitated adaptation, or assisted evolution, offers a way to augment adaptive genetic variation via artificial selection, induced hybridization, or genetic engineering. One key source of genetic variation, particularly for climatic adaptation, are the core metabolic genes encoded by the mitochondrial genome. These genes influence environmental tolerance to heat, drought, and hypoxia, but must interact intimately and co-evolve with a suite of important nuclear genes. These coadapted mitonuclear genes form some of the important reproductive barriers between species. Mitochondrial genomes can and do introgress between species in an adaptive manner, and they may co-introgress with nuclear genes important for maintaining mitonuclear compatibility. Managers should consider the relevance of mitonuclear genetic variability in conservation decision-making, including as a tool for facilitating adaptation. I propose a novel technique dubbed Conservation Mitonuclear Replacement (CmNR), which entails replacing the core metabolic machinery of a threatened species-the mitochondrial genome and key nuclear loci-with those from a closely related species or a divergent population, which may be better-adapted to climatic changes or carry a lower genetic load. The most feasible route to CmNR is to combine CRISPR-based nuclear genetic editing with mitochondrial replacement and assisted reproductive technologies. This method preserves much of an organism's phenotype and could allow populations to persist in the wild when no other suitable conservation options exist. The technique could be particularly important on mountaintops, where rising temperatures threaten an alarming number of species with almost certain extinction in the next century.
Keywords: assisted evolution; climate change; conservation; facilitated adaptation; genetic rescue; mitochondria; mitonuclear interactions.
© 2024 The Authors. Evolutionary Applications published by John Wiley & Sons Ltd.
Conflict of interest statement
The author declares no conflict of interest.
Figures
Similar articles
-
Reconciling the Mitonuclear Compatibility Species Concept with Rampant Mitochondrial Introgression.Integr Comp Biol. 2019 Oct 1;59(4):912-924. doi: 10.1093/icb/icz019. Integr Comp Biol. 2019. PMID: 30937430
-
The role of mitonuclear incompatibilities in allopatric speciation.Cell Mol Life Sci. 2022 Jan 29;79(2):103. doi: 10.1007/s00018-021-04059-3. Cell Mol Life Sci. 2022. PMID: 35091831 Free PMC article. Review.
-
Mitonuclear coevolution as the genesis of speciation and the mitochondrial DNA barcode gap.Ecol Evol. 2016 Jul 22;6(16):5831-42. doi: 10.1002/ece3.2338. eCollection 2016 Aug. Ecol Evol. 2016. PMID: 27547358 Free PMC article.
-
Assessing the fitness consequences of mitonuclear interactions in natural populations.Biol Rev Camb Philos Soc. 2019 Jun;94(3):1089-1104. doi: 10.1111/brv.12493. Epub 2018 Dec 26. Biol Rev Camb Philos Soc. 2019. PMID: 30588726 Free PMC article.
-
The on-again, off-again relationship between mitochondrial genomes and species boundaries.Mol Ecol. 2017 Apr;26(8):2212-2236. doi: 10.1111/mec.13959. Epub 2017 Jan 27. Mol Ecol. 2017. PMID: 27997046 Free PMC article. Review.
Cited by
-
An evolving roadmap: using mitochondrial physiology to help guide conservation efforts.Conserv Physiol. 2024 Sep 7;12(1):coae063. doi: 10.1093/conphys/coae063. eCollection 2024. Conserv Physiol. 2024. PMID: 39252884 Free PMC article.
-
The Complexities of Interspecies Somatic Cell Nuclear Transfer: From Biological and Molecular Insights to Future Perspectives.Int J Mol Sci. 2025 Apr 2;26(7):3310. doi: 10.3390/ijms26073310. Int J Mol Sci. 2025. PMID: 40244161 Free PMC article. Review.
-
Toward the Development of the Trojan Female Technique in Pest Insects: Male-Specific Influence of Mitochondrial Haplotype on Reproductive Output in the Seed Beetle Acanthoscelides obtectus.Evol Appl. 2024 Dec 26;17(12):e70065. doi: 10.1111/eva.70065. eCollection 2024 Dec. Evol Appl. 2024. PMID: 39726737 Free PMC article.
References
-
- Agostini, S. , Houlbrèque, F. , Biscéré, T. , Harvey, B. P. , Heitzman, J. M. , Takimoto, R. , Yamazaki, W. , Milazzo, M. , & Rodolfo‐metalpa, R. (2021). Greater mitochondrial energy production provides resistance to ocean acidification in “winning” hermatypic corals. Frontiers in Marine Science, 7, 1–11. 10.3389/fmars.2020.600836 - DOI
-
- Aguilar‐López, B. A. , Moreno‐Altamirano, M. M. B. , Dockrell, H. M. , Duchen, M. R. , & Sánchez‐García, F. J. (2020). Mitochondria: An integrative hub coordinating circadian rhythms, metabolism, the microbiome, and immunity. Frontiers in Cell and Developmental Biology, 8, 1–16. 10.3389/fcell.2020.00051 - DOI - PMC - PubMed
-
- Aitken, S. N. , & Whitlock, M. C. (2013). Assisted gene flow to facilitate local adaptation to climate change. Annual Review of Ecology, Evolution, and Systematics, 44, 367–388. 10.1146/annurev-ecolsys-110512-135747 - DOI
LinkOut - more resources
Full Text Sources

