Twelve-year review of galactosemia newborn screening in Taiwan: Evolving methods and insights
- PMID: 38469088
- PMCID: PMC10926206
- DOI: 10.1016/j.ymgmr.2024.101048
Twelve-year review of galactosemia newborn screening in Taiwan: Evolving methods and insights
Abstract
Background: Galactosemia was introduced into Taiwan's routine newborn screening (NBS) program in 1985. This study presents a 12-year experience, emphasizing disease diagnosis and screening performance.
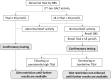
Method: NBS for galactosemia utilized dried blood spot samples taken 48-72 h post-delivery, with total galactose (TGal) level as the primary marker. Newborns with critical TGal levels were referred immediately, while those with borderline TGal underwent a recall test. GALT activity measurement was applied simultaneously as the second-tier marker. Further confirmatory tests, such as whole exome sequencing (WES), were conducted upon referral.
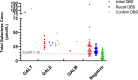
Results: From January 1st, 2011, to December 31st, 2022, 51 cases were identified from 817,906 newborns. Of these, nine individuals had persistently elevated TGal. Diagnoses included one case of GALT deficiency, one of GALM deficiency, and seven of GALE deficiencies. Notably, the classic galactosemia patient (GALT deficiency) presented with extreme high TGal and was referred to the hospital for diet management immediately. All affected patients were instructed to adopt a galactose-restricted diet. By the median age of 2.5 years, all exhibited normal development and liver function.
Conclusion: The incidence of classical galactosemia and its variants is extremely low in Taiwan. Incorporating WES into NBS has improved our ability to detect various galactosemia forms, enriching our understanding of the genetic underpinnings. While these newly discovered forms often present with milder initial elevations in TGal, specific biochemical investigations and regular monitoring are essential to understanding the long-term implications and outcomes.
Keywords: Epimerase deficiency galactosemia; GALE deficiency; Galactosemia; Newborn screening; Whole exome sequencing.
© 2024 The Authors.
Conflict of interest statement
None.
Figures
Similar articles
-
Rethinking Newborn Screening: A Case of GALM Deficiency.Int J Neonatal Screen. 2025 Apr 11;11(2):25. doi: 10.3390/ijns11020025. Int J Neonatal Screen. 2025. PMID: 40265446 Free PMC article.
-
Recommendations for newborn screening for galactokinase deficiency: A systematic review and evaluation of Dutch newborn screening data.Mol Genet Metab. 2018 May;124(1):50-56. doi: 10.1016/j.ymgme.2018.03.008. Epub 2018 Mar 21. Mol Genet Metab. 2018. PMID: 29580649
-
Galactosemia: when is it a newborn screening emergency?Mol Genet Metab. 2012 May;106(1):7-11. doi: 10.1016/j.ymgme.2012.03.007. Epub 2012 Mar 21. Mol Genet Metab. 2012. PMID: 22483615 Review.
-
The Discovery of GALM Deficiency (Type IV Galactosemia) and Newborn Screening System for Galactosemia in Japan.Int J Neonatal Screen. 2021 Oct 25;7(4):68. doi: 10.3390/ijns7040068. Int J Neonatal Screen. 2021. PMID: 34842598 Free PMC article. Review.
-
Addition of galactose-1-phosphate measurement enhances newborn screening for classical galactosemia.J Inherit Metab Dis. 2023 Mar;46(2):232-242. doi: 10.1002/jimd.12580. Epub 2022 Dec 20. J Inherit Metab Dis. 2023. PMID: 36515074
Cited by
-
Rethinking Newborn Screening: A Case of GALM Deficiency.Int J Neonatal Screen. 2025 Apr 11;11(2):25. doi: 10.3390/ijns11020025. Int J Neonatal Screen. 2025. PMID: 40265446 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

