The subdued post-boost spike-directed secondary IgG antibody response in Ugandan recipients of the Pfizer-BioNTech BNT162b2 vaccine has implications for local vaccination policies
- PMID: 38469296
- PMCID: PMC10926532
- DOI: 10.3389/fimmu.2024.1325387
The subdued post-boost spike-directed secondary IgG antibody response in Ugandan recipients of the Pfizer-BioNTech BNT162b2 vaccine has implications for local vaccination policies
Abstract
Introduction: This study aimed to delineate longitudinal antibody responses to the Pfizer-BioNTech BNT162b2 COVID-19 vaccine within the Ugandan subset of the Sub-Saharan African (SSA) demographic, filling a significant gap in global datasets.
Methods: We enrolled 48 participants and collected 320 specimens over 12 months after the primary vaccination dose. A validated enzyme-linked immunosorbent assay (ELISA) was used to quantify SARS-CoV-2-specific IgG, IgM, and IgA antibody concentrations (ng/ml) and optical densities (ODs). Statistical analyses included box plots, diverging bar graphs, and the Wilcoxon test with Bonferroni correction.
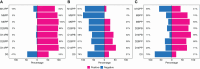
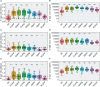
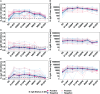
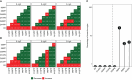
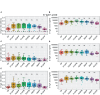
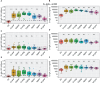
Results: We noted a robust S-IgG response within 14 days of the primary vaccine dose, which was consistent with global data. There was no significant surge in S-IgG levels after the booster dose, contrasting trends in other global populations. The S-IgM response was transient and predominantly below established thresholds for this population, which reflects its typical early emergence and rapid decline. S-IgA levels rose after the initial dose then decreased after six months, aligning with the temporal patterns of mucosal immunity. Eleven breakthrough infections were noted, and all were asymptomatic, regardless of the participants' initial S-IgG serostatus, which suggests a protective effect from vaccination.
Discussion: The Pfizer-BioNTech BNT162b2 COVID-19 vaccine elicited strong S-IgG responses in the SSA demographic. The antibody dynamics distinctly differed from global data highlighting the significance of region-specific research and the necessity for customised vaccination strategies.
Keywords: IgG IgM and IgA antibodies; Pfizer BioNTech BNT162b2 COVID-19 vaccine; Sub-Saharan African populations; Ugandan population; booster dose; breakthrough infections; longitudinal antibody responses; seropositivity classification.
Copyright © 2024 Ankunda, Katende, Oluka, Sembera, Baine, Odoch, Ejou, Kato, and The COVID-19 Immunoprofiling Team, Kaleebu and Serwanga.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Sustained S-IgG and S-IgA antibodies to Moderna's mRNA-1273 vaccine in a Sub-Saharan African cohort suggests need for booster timing reconsiderations.Front Immunol. 2024 Jan 31;15:1348905. doi: 10.3389/fimmu.2024.1348905. eCollection 2024. Front Immunol. 2024. PMID: 38357547 Free PMC article.
-
Seroprevalence and durability of antibody responses to AstraZeneca vaccination in Ugandans with prior mild or asymptomatic COVID-19: implications for vaccine policy.Front Immunol. 2023 May 2;14:1183983. doi: 10.3389/fimmu.2023.1183983. eCollection 2023. Front Immunol. 2023. PMID: 37205095 Free PMC article.
-
Real-world serological responses to extended-interval and heterologous COVID-19 mRNA vaccination in frail, older people (UNCoVER): an interim report from a prospective observational cohort study.Lancet Healthy Longev. 2022 Mar;3(3):e166-e175. doi: 10.1016/S2666-7568(22)00012-5. Epub 2022 Feb 23. Lancet Healthy Longev. 2022. PMID: 35224524 Free PMC article.
-
COVID-19 lateral flow IgG seropositivity and serum neutralising antibody responses after primary and booster vaccinations in Chile: a cross-sectional study.Lancet Microbe. 2023 Mar;4(3):e149-e158. doi: 10.1016/S2666-5247(22)00290-7. Epub 2023 Jan 27. Lancet Microbe. 2023. PMID: 36716754 Free PMC article.
-
Sustained spike-specific IgG antibodies following CoronaVac (Sinovac) vaccination in sub-Saharan Africa, but increased breakthrough infections in baseline spike-naive individuals.Front Immunol. 2023 Nov 30;14:1255676. doi: 10.3389/fimmu.2023.1255676. eCollection 2023. Front Immunol. 2023. PMID: 38098482 Free PMC article.
Cited by
-
Safety and Immunogenicity of a Modified Self-Amplifying Ribonucleic Acid (saRNA) Vaccine Encoding SARS-CoV-2 Spike Glycoprotein in SARS-CoV-2 Seronegative and Seropositive Ugandan Individuals.Vaccines (Basel). 2025 May 23;13(6):553. doi: 10.3390/vaccines13060553. Vaccines (Basel). 2025. PMID: 40573884 Free PMC article.
-
Functional antibody responses to SARS-CoV-2 variants before and after booster vaccination among adults in Ghana.Exp Biol Med (Maywood). 2025 Jul 21;250:10440. doi: 10.3389/ebm.2025.10440. eCollection 2025. Exp Biol Med (Maywood). 2025. PMID: 40761773 Free PMC article.
-
The single-dose Janssen Ad26.COV2.S COVID-19 vaccine elicited robust and persistent anti-spike IgG antibody responses in a 12-month Ugandan cohort.Front Immunol. 2024 May 8;15:1384668. doi: 10.3389/fimmu.2024.1384668. eCollection 2024. Front Immunol. 2024. PMID: 38779677 Free PMC article.
References
-
- Muir R, Metcalf T, Fourati S, Bartsch Y, Kyosiimire-Lugemwa J, Canderan G, et al. . Schistosoma mansoni infection alters the host pre-vaccination environment resulting in blunted Hepatitis B vaccination immune responses. PloS Negl Trop Dis (2023) 17:e0011089. doi: 10.1371/journal.pntd.0011089 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

