Omega-conotoxin MVIIA reduces neuropathic pain after spinal cord injury by inhibiting N-type voltage-dependent calcium channels on spinal dorsal horn
- PMID: 38469570
- PMCID: PMC10925679
- DOI: 10.3389/fnins.2024.1366829
Omega-conotoxin MVIIA reduces neuropathic pain after spinal cord injury by inhibiting N-type voltage-dependent calcium channels on spinal dorsal horn
Abstract
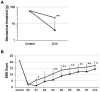
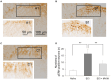
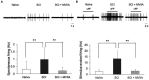
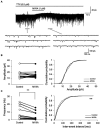
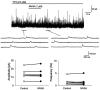
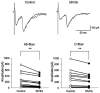
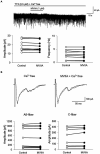
Spinal cord injury (SCI) leads to the development of neuropathic pain. Although a multitude of pathological processes contribute to SCI-induced pain, excessive intracellular calcium accumulation and voltage-gated calcium-channel upregulation play critical roles in SCI-induced pain. However, the role of calcium-channel blockers in SCI-induced pain is unknown. Omega-conotoxin MVIIA (MVIIA) is a calcium-channel blocker that selectively inhibits N-type voltage-dependent calcium channels and demonstrates neuroprotective effects. Therefore, we investigated spinal analgesic actions and cellular mechanisms underlying the analgesic effects of MVIIA in SCI. We used SCI-induced pain model rats and conducted behavioral tests, immunohistochemical analyses, and electrophysiological experiments (in vitro whole-cell patch-clamp recording and in vivo extracellular recording). A behavior study suggested intrathecal MVIIA administration in the acute phase after SCI induced analgesia for mechanical allodynia. Immunohistochemical experiments and in vivo extracellular recordings suggested that MVIIA induces analgesia in SCI-induced pain by directly inhibiting neuronal activity in the superficial spinal dorsal horn. In vitro whole-cell patch-clamp recording showed that MVIIA inhibits presynaptic N-type voltage-dependent calcium channels expressed on primary afferent Aδ-and C-fiber terminals and suppresses the presynaptic glutamate release from substantia gelatinosa in the spinal dorsal horn. In conclusion, MVIIA administration in the acute phase after SCI may induce analgesia in SCI-induced pain by inhibiting N-type voltage-dependent calcium channels on Aδ-and C-fiber terminals in the spinal dorsal horn, resulting in decreased neuronal excitability enhanced by SCI-induced pain.
Keywords: N-type voltagedependent calcium channels; neuropathic pain; omega-conotoxin MVIIA; spinal cord injury; spinal dorsal horn.
Copyright © 2024 Ohashi, Uta, Ohashi, Hoshino and Baba.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Injury-specific functional alteration of N-type voltage-gated calcium channels in synaptic transmission of primary afferent C-fibers in the rat spinal superficial dorsal horn.Eur J Pharmacol. 2016 Feb 5;772:11-21. doi: 10.1016/j.ejphar.2015.12.031. Epub 2015 Dec 19. Eur J Pharmacol. 2016. PMID: 26708163
-
Ivabradine reduces neuropathic pain after spinal cord injury by inhibiting excitatory synaptic transmission in the spinal dorsal horn.Neurosci Lett. 2025 Feb 6;848:138113. doi: 10.1016/j.neulet.2025.138113. Epub 2025 Jan 5. Neurosci Lett. 2025. PMID: 39765281
-
Spinal actions of ω-conotoxins, CVID, MVIIA and related peptides in a rat neuropathic pain model.Br J Pharmacol. 2013 Sep;170(2):245-54. doi: 10.1111/bph.12251. Br J Pharmacol. 2013. PMID: 23713957 Free PMC article.
-
Analgesic conotoxins: block and G protein-coupled receptor modulation of N-type (Ca(V) 2.2) calcium channels.Br J Pharmacol. 2012 May;166(2):486-500. doi: 10.1111/j.1476-5381.2011.01781.x. Br J Pharmacol. 2012. PMID: 22091786 Free PMC article. Review.
-
Calcium channel modulation as a target in chronic pain control.Br J Pharmacol. 2018 Jun;175(12):2173-2184. doi: 10.1111/bph.13789. Epub 2017 Apr 26. Br J Pharmacol. 2018. PMID: 28320042 Free PMC article. Review.
Cited by
-
PIONOCONUS: A PISCIVOROUS SUBGENUS OF CONUS GASTROPODS.Malacologia. 2025 Mar;67(1-2):65-116. doi: 10.4002/040.067.0104. Epub 2025 Mar 25. Malacologia. 2025. PMID: 40672043 Free PMC article.
-
Systemic antihyperalgesic effect of a novel conotoxin from Californiconus californicus in an inflammatory pain model.Front Pain Res (Lausanne). 2025 Jan 24;5:1500789. doi: 10.3389/fpain.2024.1500789. eCollection 2024. Front Pain Res (Lausanne). 2025. PMID: 39925365 Free PMC article.
-
Marine Peptides: Potential Basic Structures for the Development of Hybrid Compounds as Multitarget Therapeutics for the Treatment of Multifactorial Diseases.Int J Mol Sci. 2024 Nov 23;25(23):12601. doi: 10.3390/ijms252312601. Int J Mol Sci. 2024. PMID: 39684313 Free PMC article. Review.
References
-
- Andoh T., Uta D., Kato M., Toume K., Komatsu K., Kuraishi Y. (2017). Prophylactic administration of aucubin inhibits paclitaxel-induced mechanical allodynia via the inhibition of endoplasmic reticulum stress in peripheral Schwann cells. Biol. Pharm. Bull. 40, 473–478. doi: 10.1248/bpb.b16-00899, PMID: - DOI - PubMed
LinkOut - more resources
Full Text Sources

