Clinical Course, Laboratory Findings, and Prognosis of SARS-CoV-2 Infection in Infants up to 90 Days of Age: A Single-Center Experience and a Proposal for a Management Pathway
- PMID: 38470638
- PMCID: PMC10931066
- DOI: 10.3390/healthcare12050528
Clinical Course, Laboratory Findings, and Prognosis of SARS-CoV-2 Infection in Infants up to 90 Days of Age: A Single-Center Experience and a Proposal for a Management Pathway
Abstract
Aim: To provide a comprehensive description of the clinical features, biochemical characteristics, and outcomes of infants up to 90 days old with COVID-19. Moreover, to assess the severity of the disease and propose an effective management pathway.
Methods: Retrospective single-center study spanning three years. Patient data includes age, sex, symptoms, comorbidities, blood and urine test results, cultures, admission, length of stay, therapies, intensive care unit admission, and mortality.
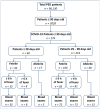
Results: A total of 274 patients were enrolled in the study, comprising 55% males. Among them, 60 patients (22%) were under the age of 29 days, while 214 (78%) fell within the 29 to 90 days age range. The overall incidence of SARS-CoV-2 infections was 0.28 per 10,000 Pediatric Emergency Department admissions. Blood inflammatory markers showed no significant abnormalities, and there were no recorded instances of positive blood cultures. Less than 1% of infants showed urinary tract infections with positive urine cultures, and 1.5% of patients had a concurrent RSV infection. Hospitalization rates were 83% for neonates and 67% for infants, with a median length of stay (LOS) of 48 h for both age groups. None of the patients required admission to the Pediatric or Neonatal Intensive Care Unit, and only one required High Flow Nasal Cannula (HFNC). No secondary serious bacterial infections were observed, and all hospitalized patients were discharged without short-term sequelae. No deaths were reported.
Discussion and conclusions: Infants with COVID-19 generally exhibit milder or asymptomatic forms of the disease, making home management a viable option in most cases. Blood tests, indicative of a mild inflammatory response, are recommended primarily for children showing symptoms of illness. Hospitalization precautions for infants without apparent illness or comorbidities are deemed unnecessary. Given the evolving nature of experiences with COVID-19 in infants, maintaining a high level of clinical suspicion remains imperative.
Keywords: COVID-19 epidemiology; clinical severity; febrile infant; pediatric emergency department; prognosis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Safety and Efficacy of Imatinib for Hospitalized Adults with COVID-19: A structured summary of a study protocol for a randomised controlled trial.Trials. 2020 Oct 28;21(1):897. doi: 10.1186/s13063-020-04819-9. Trials. 2020. PMID: 33115543 Free PMC article.
-
Testing the efficacy and safety of BIO101, for the prevention of respiratory deterioration, in patients with COVID-19 pneumonia (COVA study): a structured summary of a study protocol for a randomised controlled trial.Trials. 2021 Jan 11;22(1):42. doi: 10.1186/s13063-020-04998-5. Trials. 2021. PMID: 33430924 Free PMC article.
-
Fever without source as the first manifestation of SARS-CoV-2 infection in infants less than 90 days old.Eur J Pediatr. 2021 Jul;180(7):2099-2106. doi: 10.1007/s00431-021-03973-9. Epub 2021 Feb 19. Eur J Pediatr. 2021. PMID: 33606120 Free PMC article.
-
Community-Onset Severe Acute Respiratory Syndrome Coronavirus 2 Infection in Young Infants: A Systematic Review.J Pediatr. 2021 Jan;228:94-100.e3. doi: 10.1016/j.jpeds.2020.09.008. Epub 2020 Sep 8. J Pediatr. 2021. PMID: 32910943 Free PMC article.
-
Multisystem inflammatory syndrome in neonates (MIS-N): a systematic review.Eur J Pediatr. 2023 May;182(5):2283-2298. doi: 10.1007/s00431-023-04906-4. Epub 2023 Mar 6. Eur J Pediatr. 2023. PMID: 36877274 Free PMC article.
References
-
- Venturini E., Montagnani C., Garazzino S., Donà D., Pierantoni L., Vecchio A.L., Nicolini G., Bianchini S., Krzysztofiak A., Galli L., et al. Treatment of children with COVID-19: Position paper of the Italian Society of Pediatric Infectious Disease. Ital. J. Pediatr. 2020;46:139. doi: 10.1186/s13052-020-00900-w. - DOI - PMC - PubMed
-
- Gale C., A Quigley M., Placzek A., Knight M., Ladhani S., Draper E.S., Sharkey D., Doherty C., Mactier H., Kurinczuk J.J. Characteristics and outcomes of neonatal SARS-CoV-2 infection in the UK: A prospective national cohort study using active surveillance. Lancet Child Adolesc. Health. 2021;5:113–121. doi: 10.1016/S2352-4642(20)30342-4. - DOI - PMC - PubMed
-
- Kanburoglu M.K., Tayman C., Oncel M.Y., Akin I.M., Can E., Demir N., Arayici S., Baser D.O., Caner I., Memisoglu A., et al. A Multicentered Study on Epidemiologic and Clinical Characteristics of 37 Neonates With Community-acquired COVID-19. Pediatr. Infect. Dis. J. 2020;39:e297–e302. doi: 10.1097/INF.0000000000002862. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous