Metalloradical Catalysis: General Approach for Controlling Reactivity and Selectivity of Homolytic Radical Reactions
- PMID: 38472114
- PMCID: PMC11097140
- DOI: 10.1002/anie.202320243
Metalloradical Catalysis: General Approach for Controlling Reactivity and Selectivity of Homolytic Radical Reactions
Abstract
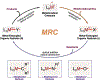
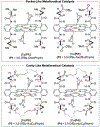
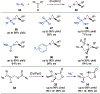
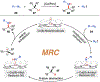
Since Friedrich Wöhler's groundbreaking synthesis of urea in 1828, organic synthesis over the past two centuries has predominantly relied on the exploration and utilization of chemical reactions rooted in two-electron heterolytic ionic chemistry. While one-electron homolytic radical chemistry is both rich in fundamental reactivities and attractive with practical advantages, the synthetic application of radical reactions has been long hampered by the formidable challenges associated with the control over reactivity and selectivity of high-energy radical intermediates. To fully harness the untapped potential of radical chemistry for organic synthesis, there is a pressing need to formulate radically different concepts and broadly applicable strategies to address these outstanding issues. In pursuit of this objective, researchers have been actively developing metalloradical catalysis (MRC) as a comprehensive framework to guide the design of general approaches for controlling over reactivity and stereoselectivity of homolytic radical reactions. Essentially, MRC exploits the metal-centered radicals present in open-shell metal complexes as one-electron catalysts for homolytic activation of substrates to generate metal-entangled organic radicals as the key intermediates to govern the reaction pathway and stereochemical course of subsequent catalytic radical processes. Different from the conventional two-electron catalysis by transition metal complexes, MRC operates through one-electron chemistry utilizing stepwise radical mechanisms.
Keywords: C−H functionalization; aziridination; cyclopropanation; metalloradical catalysis; radical reaction.
© 2024 The Authors. Angewandte Chemie International Edition published by Wiley-VCH GmbH.
Conflict of interest statement
Conflict of Interest
The authors declare no conflict of interest.
Figures

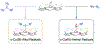

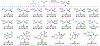
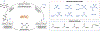

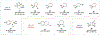
Similar articles
-
Iron(III)-based metalloradical catalysis for asymmetric cyclopropanation via a stepwise radical mechanism.Nat Chem. 2023 Nov;15(11):1569-1580. doi: 10.1038/s41557-023-01317-8. Epub 2023 Sep 7. Nat Chem. 2023. PMID: 37679462 Free PMC article.
-
When Light Meets Nitrogen-Centered Radicals: From Reagents to Catalysts.Acc Chem Res. 2020 May 19;53(5):1066-1083. doi: 10.1021/acs.accounts.0c00090. Epub 2020 Apr 14. Acc Chem Res. 2020. PMID: 32286794
-
Nitrene Radical Intermediates in Catalytic Synthesis.Chemistry. 2017 Oct 9;23(56):13819-13829. doi: 10.1002/chem.201702537. Epub 2017 Sep 14. Chemistry. 2017. PMID: 28675476 Free PMC article.
-
Radicals in transition metal catalyzed reactions? transition metal catalyzed radical reactions? a fruitful interplay anyway: part 1. Radical catalysis by group 4 to group 7 elements.Top Curr Chem. 2012;320:121-89. doi: 10.1007/128_2011_261. Top Curr Chem. 2012. PMID: 22025066 Review.
-
Radical approaches for C(sp3)-N bond cleavage in deaminative transformations.Chem Commun (Camb). 2025 May 8;61(39):6997-7008. doi: 10.1039/d5cc01476j. Chem Commun (Camb). 2025. PMID: 40265294 Review.
Cited by
-
Cyano group translocation to alkenyl C(sp2)-H site by radical cation catalysis.Nat Commun. 2025 Aug 6;16(1):7251. doi: 10.1038/s41467-025-62585-1. Nat Commun. 2025. PMID: 40769981 Free PMC article.
-
Asymmetric C-H Amination via Fe(III)-Metalloradical Catalysis Featuring α-Fe(IV)-Aminyl Radicals as Key Intermediates.J Am Chem Soc. 2025 Jul 9;147(27):24001-24013. doi: 10.1021/jacs.5c07473. Epub 2025 Jun 19. J Am Chem Soc. 2025. PMID: 40534495
-
Building Catalytic Reactions One Electron at a Time.Acc Chem Res. 2024 Oct 15;57(20):3068-3078. doi: 10.1021/acs.accounts.4c00515. Epub 2024 Sep 24. Acc Chem Res. 2024. PMID: 39317431
-
Preassembly-Controlled Radical Recombination at Bismuth: Decarboxylative C─N Coupling with Sulfonamides.Chemistry. 2025 May 19;31(28):e202500396. doi: 10.1002/chem.202500396. Epub 2025 Apr 21. Chemistry. 2025. PMID: 40192563 Free PMC article.
-
Catalytic Metalloradical System for Radical 1,6-C(sp3)-H Amination with Concurrent Control of Site-, Chemo-, and Enantio-selectivity.J Am Chem Soc. 2025 May 7;147(18):15755-15766. doi: 10.1021/jacs.5c03259. Epub 2025 Apr 22. J Am Chem Soc. 2025. PMID: 40263670
References
-
- Curran DP, Porter NA, Giese B, Stereochemistry of Radical Reactions: Concepts, Guidelines, and Synthetic Applications, John Wiley & Sons, Weinheim; New York, 2008.
-
- Mondal S, Dumur F, Gigmes D, Sibi MP, Bertrand MP, Nechab M, Chem. Rev 2022, 122, 5842–5976. - PubMed
-
- Lu HJ, Zhang XP, Chem. Soc. Rev 2011, 40, 1899–1909. - PubMed
-
- Wang X, Zhang XP, Catalytic Radical Approach for Selective Carbene Transfers via Cobalt(II)-Based Metalloradical Catalysis, in Transition Metal-Catalyzed Carbene Transformations, Wiley, 2022, pp. 25–66.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

