Intrinsic Lipid Curvature and Bilayer Elasticity as Regulators of Channel Function: A Comparative Single-Molecule Study
- PMID: 38474005
- PMCID: PMC10931550
- DOI: 10.3390/ijms25052758
Intrinsic Lipid Curvature and Bilayer Elasticity as Regulators of Channel Function: A Comparative Single-Molecule Study
Abstract
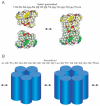
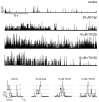
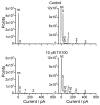
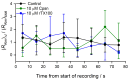
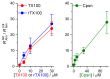
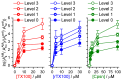
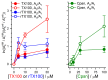
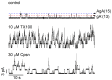
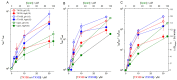
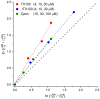
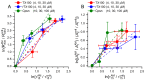
Perturbations in bilayer material properties (thickness, lipid intrinsic curvature and elastic moduli) modulate the free energy difference between different membrane protein conformations, thereby leading to changes in the conformational preferences of bilayer-spanning proteins. To further explore the relative importance of curvature and elasticity in determining the changes in bilayer properties that underlie the modulation of channel function, we investigated how the micelle-forming amphiphiles Triton X-100, reduced Triton X-100 and the HII lipid phase promoter capsaicin modulate the function of alamethicin and gramicidin channels. Whether the amphiphile-induced changes in intrinsic curvature were negative or positive, amphiphile addition increased gramicidin channel appearance rates and lifetimes and stabilized the higher conductance states in alamethicin channels. When the intrinsic curvature was modulated by altering phospholipid head group interactions, however, maneuvers that promote a negative-going curvature stabilized the higher conductance states in alamethicin channels but destabilized gramicidin channels. Using gramicidin channels of different lengths to probe for changes in bilayer elasticity, we found that amphiphile adsorption increases bilayer elasticity, whereas altering head group interactions does not. We draw the following conclusions: first, confirming previous studies, both alamethicin and gramicidin channels are modulated by changes in lipid bilayer material properties, the changes occurring in parallel yet differing dependent on the property that is being changed; second, isolated, negative-going changes in curvature stabilize the higher current levels in alamethicin channels and destabilize gramicidin channels; third, increases in bilayer elasticity stabilize the higher current levels in alamethicin channels and stabilize gramicidin channels; and fourth, the energetic consequences of changes in elasticity tend to dominate over changes in curvature.
Keywords: alamethicin channel; amphiphiles; bilayer-mediated regulation; elasticity; gramicidin channel; lipid intrinsic curvature.
Conflict of interest statement
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analysis or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
Similar articles
-
Regulation of sodium channel function by bilayer elasticity: the importance of hydrophobic coupling. Effects of Micelle-forming amphiphiles and cholesterol.J Gen Physiol. 2004 May;123(5):599-621. doi: 10.1085/jgp.200308996. J Gen Physiol. 2004. PMID: 15111647 Free PMC article.
-
Single-molecule methods for monitoring changes in bilayer elastic properties.Methods Mol Biol. 2007;400:543-70. doi: 10.1007/978-1-59745-519-0_37. Methods Mol Biol. 2007. PMID: 17951759 Review.
-
Amphiphile regulation of ion channel function by changes in the bilayer spring constant.Proc Natl Acad Sci U S A. 2010 Aug 31;107(35):15427-30. doi: 10.1073/pnas.1007455107. Epub 2010 Aug 16. Proc Natl Acad Sci U S A. 2010. PMID: 20713738 Free PMC article.
-
Interactions of drugs and amphiphiles with membranes: modulation of lipid bilayer elastic properties by changes in acyl chain unsaturation and protonation.Faraday Discuss. 2013;161:461-80; discussion 563-89. doi: 10.1039/c2fd20092a. Faraday Discuss. 2013. PMID: 23805753 Free PMC article.
-
Lipid bilayer regulation of membrane protein function: gramicidin channels as molecular force probes.J R Soc Interface. 2010 Mar 6;7(44):373-95. doi: 10.1098/rsif.2009.0443. Epub 2009 Nov 25. J R Soc Interface. 2010. PMID: 19940001 Free PMC article. Review.
Cited by
-
Amphiphiles capsaicin and triton X-100 regulate the chemotherapy drug colchicine's membrane adsorption and ion pore formation potency.Saudi J Biol Sci. 2021 May;28(5):3100-3109. doi: 10.1016/j.sjbs.2021.02.054. Epub 2021 Feb 21. Saudi J Biol Sci. 2021. PMID: 34025185 Free PMC article.
-
The Antimicrobial Peptide Gramicidin S Enhances Membrane Adsorption and Ion Pore Formation Potency of Chemotherapy Drugs in Lipid Bilayers.Membranes (Basel). 2021 Mar 30;11(4):247. doi: 10.3390/membranes11040247. Membranes (Basel). 2021. PMID: 33808204 Free PMC article.
-
Examining changes in gramicidin current induced by endocannabinoids.PLoS One. 2025 Aug 18;20(8):e0313903. doi: 10.1371/journal.pone.0313903. eCollection 2025. PLoS One. 2025. PMID: 40825012 Free PMC article.
-
Endocannabinoid regulation of inward rectifier potassium (Kir) channels.Front Pharmacol. 2024 Aug 26;15:1439767. doi: 10.3389/fphar.2024.1439767. eCollection 2024. Front Pharmacol. 2024. PMID: 39253376 Free PMC article.
References
-
- Bienvenüe A., Marie J.S. Modulation of protein function by lipids. Curr. Top. Membr. 1994;40:319–354.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

