Exploring Individual Variability in Drug-Induced Liver Injury (DILI) Responses through Metabolomic Analysis
- PMID: 38474249
- PMCID: PMC10932304
- DOI: 10.3390/ijms25053003
Exploring Individual Variability in Drug-Induced Liver Injury (DILI) Responses through Metabolomic Analysis
Abstract
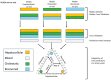
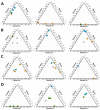
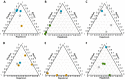
Drug-induced liver injury (DILI) is a serious adverse hepatic event presenting diagnostic and prognostic challenges. The clinical categorization of DILI into hepatocellular, cholestatic, or mixed phenotype is based on serum alanine aminotransferase (ALT) and alkaline phosphatase (ALP) values; however, this classification may not capture the full spectrum of DILI subtypes. With this aim, we explored the utility of assessing changes in the plasma metabolomic profiles of 79 DILI patients assessed by the RUCAM (Roussel Uclaf Causality Assessment Method) score to better characterize this condition and compare results obtained with the standard clinical characterization. Through the identification of various metabolites in the plasma (including free and conjugated bile acids and glycerophospholipids), and the integration of this information into predictive models, we were able to evaluate the extent of the hepatocellular or cholestatic phenotype and to assign a numeric value with the contribution of each specific DILI sub-phenotype into the patient's general condition. Additionally, our results showed that metabolomic analysis enabled the monitoring of DILI variability responses to the same drug, the transitions between sub-phenotypes during disease progression, and identified a spectrum of residual DILI metabolic features, which can be overlooked using standard clinical diagnosis during patient follow-up.
Keywords: DILI; biomarkers; cholestasis; hepatocellular damage; hepatotoxicity; idiosyncratic DILI; metabolomics; updated RUCAM.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


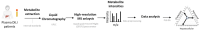
Similar articles
-
Metabolomic analysis to discriminate drug-induced liver injury (DILI) phenotypes.Arch Toxicol. 2021 Sep;95(9):3049-3062. doi: 10.1007/s00204-021-03114-z. Epub 2021 Jul 17. Arch Toxicol. 2021. PMID: 34274980 Free PMC article.
-
Utility of Lymphocyte Transformation Test for Assisting Updated Roussel Uclaf Causality Assessment Method in Drug-Induced Liver Injury: A Case-Control Study.Front Pharmacol. 2022 Mar 10;13:819589. doi: 10.3389/fphar.2022.819589. eCollection 2022. Front Pharmacol. 2022. PMID: 35370653 Free PMC article.
-
Drug Induced Liver Injury: Can Biomarkers Assist RUCAM in Causality Assessment?Int J Mol Sci. 2017 Apr 11;18(4):803. doi: 10.3390/ijms18040803. Int J Mol Sci. 2017. PMID: 28398242 Free PMC article. Review.
-
Advances in Idiosyncratic Drug-Induced Liver Injury Issues: New Clinical and Mechanistic Analysis Due to Roussel Uclaf Causality Assessment Method Use.Int J Mol Sci. 2023 Jun 29;24(13):10855. doi: 10.3390/ijms241310855. Int J Mol Sci. 2023. PMID: 37446036 Free PMC article. Review.
-
Identification of Distinguishing Features of Drug-Induced Liver Injury and Liver Injury Associated With Sepsis.Liver Int. 2025 May;45(5):e70090. doi: 10.1111/liv.70090. Liver Int. 2025. PMID: 40257426
References
-
- Andrade R.J., Lucena M.I., Kaplowitz N., García-Muņoz B., Borraz Y., Pachkoria K., García-Cortés M., Fernández M.C., Pelaez G., Rodrigo L., et al. Outcome of acute idiosyncratic drug-induced liver injury: Long-term follow-up in a hepatotoxicity registry. Hepatology. 2006;44:1581–1588. doi: 10.1002/hep.21424. - DOI - PubMed
-
- Andrade R.J., Lucena M.I., Fernández M.C., Pelaez G., Pachkoria K., García-Ruiz E., García-Muñoz B., González-Grande R., Pizarro A., Durán J.A., et al. Drug-induced liver injury: An analysis of 461 incidences submitted to the Spanish registry over a 10-year period. Gastroenterology. 2005;129:512–521. doi: 10.1016/j.gastro.2005.05.006. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

