Cholangiocarcinoma Malignant Traits Are Promoted by Schwann Cells through TGFβ Signaling in a Model of Perineural Invasion
- PMID: 38474330
- PMCID: PMC10930666
- DOI: 10.3390/cells13050366
Cholangiocarcinoma Malignant Traits Are Promoted by Schwann Cells through TGFβ Signaling in a Model of Perineural Invasion
Abstract
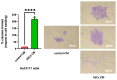
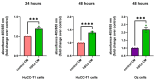
The term cholangiocarcinoma (CCA) defines a class of epithelial malignancies originating from bile ducts. Although it has been demonstrated that CCA patients with perineural invasion (PNI) have a worse prognosis, the biological features of this phenomenon are yet unclear. Our data show that in human intrahepatic CCA specimens with documented PNI, nerve-infiltrating CCA cells display positivity of the epithelial marker cytokeratin 7, lower with respect to the rest of the tumor mass. In an in vitro 3D model, CCA cells move towards a peripheral nerve explant allowing contact with Schwann cells (SCs) emerging from the nerve. Here, we show that SCs produce soluble factors that favor the migration, invasion, survival and proliferation of CCA cells in vitro. This effect is accompanied by a cadherin switch, suggestive of an epithelial-mesenchymal transition. The influence of SCs in promoting the ability of CCA cells to migrate and invade the extracellular matrix is hampered by a specific TGFβ receptor 1 (TGFBR1) antagonist. Differential proteomic data indicate that the exposure of CCA cells to SC secreted factors induces the upregulation of key oncogenes and the concomitant downregulation of some tumor suppressors. Taken together, these data concur in identifying SCs as possible promoters of a more aggressive CCA phenotype, ascribing a central role to TGFβ signaling in regulating this process.
Keywords: EMT; Schwann cells; TGFβ; bile duct cancer.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures







Similar articles
-
Promotion of perineural invasion of cholangiocarcinoma by Schwann cells via nerve growth factor.J Gastrointest Oncol. 2024 Jun 30;15(3):1198-1213. doi: 10.21037/jgo-24-309. Epub 2024 Jun 27. J Gastrointest Oncol. 2024. PMID: 38989424 Free PMC article.
-
EGF/EGFR axis contributes to the progression of cholangiocarcinoma through the induction of an epithelial-mesenchymal transition.J Hepatol. 2014 Aug;61(2):325-32. doi: 10.1016/j.jhep.2014.03.033. Epub 2014 Apr 3. J Hepatol. 2014. PMID: 24704591
-
Loss of E-cadherin promotes migration and invasion of cholangiocarcinoma cells and serves as a potential marker of metastasis.Tumour Biol. 2014 Sep;35(9):8645-52. doi: 10.1007/s13277-014-2087-6. Epub 2014 May 28. Tumour Biol. 2014. PMID: 24867095
-
The role of tumor-infiltrating lymphocytes in cholangiocarcinoma.J Exp Clin Cancer Res. 2022 Apr 7;41(1):127. doi: 10.1186/s13046-022-02340-2. J Exp Clin Cancer Res. 2022. PMID: 35392957 Free PMC article. Review.
-
Wnt/β-catenin signaling as an emerging potential key pharmacological target in cholangiocarcinoma.Biosci Rep. 2020 Mar 27;40(3):BSR20193353. doi: 10.1042/BSR20193353. Biosci Rep. 2020. PMID: 32140709 Free PMC article. Review.
Cited by
-
New Relevant Evidence in Cholangiocarcinoma Biology and Characterization.Cancers (Basel). 2024 Dec 19;16(24):4239. doi: 10.3390/cancers16244239. Cancers (Basel). 2024. PMID: 39766138 Free PMC article. Review.
-
The Emerging Role of Schwann Cells in the Tumor Immune Microenvironment and Its Potential Clinical Application.Int J Mol Sci. 2024 Dec 23;25(24):13722. doi: 10.3390/ijms252413722. Int J Mol Sci. 2024. PMID: 39769484 Free PMC article. Review.
-
Unraveling the role of perineural invasion in cancer progression across multiple tumor types.Med Oncol. 2025 Jun 26;42(8):283. doi: 10.1007/s12032-025-02855-6. Med Oncol. 2025. PMID: 40569482 Review.
-
An oxidative stress related gene signature predicts prognosis in cholangiocarcinoma.Discov Oncol. 2025 Aug 18;16(1):1576. doi: 10.1007/s12672-025-03434-x. Discov Oncol. 2025. PMID: 40824414 Free PMC article.
-
Roles of Peripheral Nerves in Tumor Initiation and Progression.Int J Mol Sci. 2025 Jul 22;26(15):7064. doi: 10.3390/ijms26157064. Int J Mol Sci. 2025. PMID: 40806192 Free PMC article. Review.
References
-
- Montal R., Sia D., Montironi C., Leow W.Q., Esteban-Fabró R., Pinyol R., Torres-Martin M., Bassaganyas L., Moeini A., Peix J., et al. Molecular classification and therapeutic targets in extrahepatic cholangiocarcinoma. J. Hepatol. 2020;73:315–327. doi: 10.1016/j.jhep.2020.03.008. - DOI - PMC - PubMed
-
- Tan X., Sivakumar S., Bednarsch J., Wiltberger G., Kather J.N., Niehues J., de Vos-Geelen J., Valkenburg-van Iersel L., Kintsler S., Roeth A., et al. Nerve fibers in the tumor microenvironment in neurotropic cancer-pancreatic cancer and cholangiocarcinoma. Oncogene. 2021;40:899–908. doi: 10.1038/s41388-020-01578-4. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

