CM1, a Chrysin Derivative, Protects from Endotoxin-Induced Lethal Shock by Regulating the Excessive Activation of Inflammatory Responses
- PMID: 38474770
- PMCID: PMC10934210
- DOI: 10.3390/nu16050641
CM1, a Chrysin Derivative, Protects from Endotoxin-Induced Lethal Shock by Regulating the Excessive Activation of Inflammatory Responses
Abstract
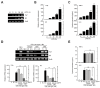
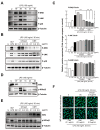
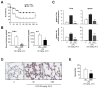
Sepsis, a leading cause of death worldwide, is a harmful inflammatory condition that is primarily caused by an endotoxin released by Gram-negative bacteria. Effective targeted therapeutic strategies for sepsis are lacking. In this study, using an in vitro and in vivo mouse model, we demonstrated that CM1, a derivative of the natural polyphenol chrysin, exerts an anti-inflammatory effect by inducing the expression of the ubiquitin-editing protein TNFAIP3 and the NAD-dependent deacetylase sirtuin 1 (SIRT1). Interestingly, CM1 attenuated the Toll-like receptor 4 (TLR4)-induced production of inflammatory cytokines by inhibiting the extracellular-signal-regulated kinase (ERK)/MAPK and nuclear factor kappa B (NF-κB) signalling pathways. In addition, CM1 induced the expression of TNFAIP3 and SIRT1 on TLR4-stimulated primary macrophages; however, the anti-inflammatory effect of CM1 was abolished by the siRNA-mediated silencing of TNFAPI3 or by the genetic or pharmacologic inhibition of SIRT1. Importantly, intravenous administration of CM1 resulted in decreased susceptibility to endotoxin-induced sepsis, thereby attenuating the production of pro-inflammatory cytokines and neutrophil infiltration into the lung compared to control mice. Collectively, these findings demonstrate that CM1 has therapeutic potential for diverse inflammatory diseases, including sepsis.
Keywords: CM1; TNFAIP3; Toll-like receptor 4; inflammation; sepsis; sirtuin 1.
Conflict of interest statement
The authors have no financial conflict of interests.
Figures




References
-
- Matera G., Quirino A., Peronace C., Settembre P., Marano V., Loria M.T., Marascio N., Galati L., Barreca G.S., Giancotti A., et al. Soluble CD14 Subtype-A New Biomarker in Predicting the Outcome of Critically Ill Septic Patients. Am. J. Med. Sci. 2017;353:543–551. doi: 10.1016/j.amjms.2017.03.036. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

