The characterization of CellROX™ probes could be a crucial factor in ram sperm quality assessment
- PMID: 38476170
- PMCID: PMC10927726
- DOI: 10.3389/fvets.2024.1342808
The characterization of CellROX™ probes could be a crucial factor in ram sperm quality assessment
Abstract
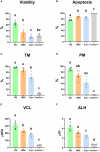
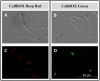
Several authors have demonstrated that low levels of reactive oxygen species (ROS) are necessary for the physiological functions of sperm, such as capacitation, hyperactivation, acrosomal reaction and fertilization. However, high levels of ROS are associated with oxidative stress and detrimental effects on fertility. Consequently, deep characterization of ROS presence using different fluorescent probes could be crucial. In this sense, the study of intracellular ROS localization and the relationships between ROS and other conventional parameters could improve the characterization of sperm quality for semen preservation protocols in rams. In this work, a multiparametric study was carried out by analyzing four experimental groups of ram sperm with different initial qualities: fresh semen (from both breeding and nonbreeding seasons), frozen-thawed semen and, a positive control group treated with hydrogen peroxide (300 μM) as a marker of extreme damage. Sperm analyses, including viability, apoptosis, lipid peroxidation, motility and kinetic parameters, were applied to compare several experimental groups with different sperm qualities. After that, the signals from two different ROS probes: CellROX™ Deep Red (CRDR) and Green (CRG), were examined by flow cytometry (percentage of cells that express ROS) and fluorescence microscopy (intracellular ROS location). Comparing conventional parameters, fresh samples from the breeding season showed the highest sperm quality, while the positive control samples showed the worst sperm quality. Concerning the ROS probes, the CRDR levels were higher in fresh samples from the breeding season than in the positive control and cryopreserved samples. Surprisingly, CRG presented its highest level (P < 0.05) in the positive control group treated with peroxide by flow cytometry. CRDR and CRG presented opposite labeling patterns that were corroborated by fluorescence microscopy, which determined that the probes localized in different parts of sperm. CRDR was found in the sperm mitochondrial region, while CRG was observed in the cell nucleus, suggesting that ROS localization is an important factor. Finally, our study indicates that CRDR is correlated with proper viability and sperm motility, and could be associated with high mitochondrial activity, while CRG is associated with sperm damage.
Keywords: CellROX probes; lipid peroxidation; ovine; oxidative stress; reactive oxygen species; sperm.
Copyright © 2024 Palacin-Martinez, Anel-Lopez, Alvarez, Neila-Montero, Montes-Garrido, Soriano-Úbeda, de Paz, Anel and Riesco.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
The Effect of Astaxanthin on Motility, Viability, Reactive Oxygen Species, Apoptosis, and Lipid Peroxidation of Human Spermatozoa During the Freezing-Thawing Process.Biopreserv Biobank. 2022 Aug;20(4):367-373. doi: 10.1089/bio.2021.0112. Biopreserv Biobank. 2022. PMID: 35984938
-
Efficiency of CellROX deep red® and CellROX orange® fluorescent probes in identifying reactive oxygen species in sperm samples from high and low fertility bulls.Anim Biotechnol. 2021 Feb;32(1):77-83. doi: 10.1080/10495398.2019.1654485. Epub 2019 Aug 19. Anim Biotechnol. 2021. PMID: 31424334
-
Effects from aging on semen quality of fresh and cryopreserved semen in Labrador Retrievers.Theriogenology. 2019 Jul 1;132:164-171. doi: 10.1016/j.theriogenology.2019.04.013. Epub 2019 Apr 17. Theriogenology. 2019. PMID: 31029847
-
Lipid peroxidation and generation of hydrogen peroxide in frozen-thawed ram semen cryopreserved in extenders with antioxidants.Anim Reprod Sci. 2010 Oct;122(1-2):118-23. doi: 10.1016/j.anireprosci.2010.08.004. Epub 2010 Aug 11. Anim Reprod Sci. 2010. PMID: 20813469
-
Influence of reactive oxygen species on human sperm functions and fertilizing capacity including therapeutical approaches.Arch Gynecol Obstet. 2013 Jul;288(1):191-9. doi: 10.1007/s00404-013-2801-4. Epub 2013 Mar 30. Arch Gynecol Obstet. 2013. PMID: 23543240 Review.
Cited by
-
Phytochemical Profiling of Processed Açaí Pulp (Euterpe oleracea) Through Mass Spectrometry and Its Protective Effects Against Oxidative Stress in Cardiomyocytes and Rats.Antioxidants (Basel). 2025 May 27;14(6):642. doi: 10.3390/antiox14060642. Antioxidants (Basel). 2025. PMID: 40563277 Free PMC article.
-
Mitoregulin Promotes Cell Cycle Progression in Non-Small Cell Lung Cancer Cells.Int J Mol Sci. 2025 Feb 24;26(5):1939. doi: 10.3390/ijms26051939. Int J Mol Sci. 2025. PMID: 40076565 Free PMC article.
-
Immune suppressive activities of low-density neutrophils in sepsis and potential use as a novel biomarker of sepsis-induced immune suppression.Sci Rep. 2025 Mar 19;15(1):9458. doi: 10.1038/s41598-025-92417-7. Sci Rep. 2025. PMID: 40108283 Free PMC article.
References
LinkOut - more resources
Full Text Sources

