Prevalence of SARS-CoV-2 infections among Swedish healthcare workers on duty in December 2023
- PMID: 38476745
- PMCID: PMC10928269
- DOI: 10.1016/j.lanepe.2024.100872
Prevalence of SARS-CoV-2 infections among Swedish healthcare workers on duty in December 2023
Conflict of interest statement
All authors declare no competing interests.
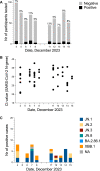
Figures
References
-
- Control ECfDPa SARS-CoV-2 variants of concern as of 15 December 2023. 2023. https://www.ecdc.europa.eu/en/covid-19/variants-concern
-
- Link-Gelles R., Ciesla A.A., Fleming-Dutra K.E., et al. Effectiveness of bivalent mRNA vaccines in preventing symptomatic SARS-CoV-2 infection — increasing community access to testing program, United States, September–November 2022. MMWR Morb Mortal Wkly Rep. 2022;71(1526–1530):1526–1530. doi: 10.15585/mmwr.mm7148e1. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

