Advancing rare disease treatment: EMA's decade-long insights into engineered adoptive cell therapy for rare cancers and orphan designation
- PMID: 38480914
- PMCID: PMC11257961
- DOI: 10.1038/s41434-024-00446-0
Advancing rare disease treatment: EMA's decade-long insights into engineered adoptive cell therapy for rare cancers and orphan designation
Abstract
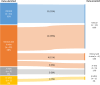
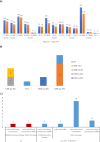
Adoptive cell therapy (ACT), particularly chimeric antigen receptor (CAR)-T cell therapy, has emerged as a promising approach for targeting and treating rare oncological conditions. The orphan medicinal product designation by the European Union (EU) plays a crucial role in promoting development of medicines for rare conditions according to the EU Orphan Regulation.This regulatory landscape analysis examines the evolution, regulatory challenges, and clinical outcomes of genetically engineered ACT, with a focus on CAR-T cell therapies, based on the European Medicines Agency's Committee for Orphan Medicinal Products review of applications evaluated for orphan designation and maintenance of the status over a 10-year period. In total, 30 of 36 applications were granted an orphan status, and 14 subsequently applied for maintenance of the status at time of marketing authorisation or extension of indication. Most of the products were autologous cell therapies using a lentiviral vector and were developed for the treatment of rare haematological B-cell malignancies. The findings revealed that 80% (29/36) of the submissions for orphan designation were supported by preliminary clinical data showing a potential efficacy of the candidate products and an added clinical benefit over currently authorised medicines for the proposed orphan condition. Notably, in 89% (32/36) of the cases significant benefit of the new products was accepted based on a clinically relevant advantage over existing therapies. Twelve of fourteen submissions reviewed for maintenance of the status at time of marketing authorisation or extension of indication demonstrated significant benefit of the products over existing satisfactory methods of treatment within the approved therapeutic indications, but one of the applications was withdrawn during the regulatory evaluation.This article summarises the key findings related to the use of engineered ACT, primarily CAR-T cell therapies, in targeting and treating rare cancers in the EU. It emphasises the importance of use of clinical data in supporting medical plausibility and significant benefit at the stage of orphan designation and highlights the high success rate for these products in obtaining initial orphan designations and subsequent maintaining the status at the time of marketing authorisation or extension of indication.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


References
-
- Grupp SA, June CH. Adoptive cellular therapy. Curr Top Microbiol Immunol. 2011;344:149–72. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

