Dynamic surveillance of lymphocyte subsets in patients with non-small cell lung cancer during chemotherapy or combination immunotherapy for early prediction of efficacy
- PMID: 38482008
- PMCID: PMC10933068
- DOI: 10.3389/fimmu.2024.1316778
Dynamic surveillance of lymphocyte subsets in patients with non-small cell lung cancer during chemotherapy or combination immunotherapy for early prediction of efficacy
Abstract
Background: Non-small cell lung cancer (NSCLC) remains the leading cause of cancer-related deaths worldwide. Lymphocytes are the primary executors of the immune system and play essential roles in tumorigenesis and development. We investigated the dynamic changes in peripheral blood lymphocyte subsets to predict the efficacy of chemotherapy or combination immunotherapy in NSCLC.
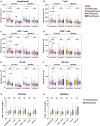
Methods: This retrospective study collected data from 81 patients with NSCLC who received treatments at the First Affiliated Hospital of Zhengzhou University from May 2021 to May 2023. Patients were divided into response and non-response groups, chemotherapy and combination immunotherapy groups, and first-line and multiline groups. We analyzed the absolute counts of each lymphocyte subset in the peripheral blood at baseline and after each treatment cycle. Within-group and between-group differences were analyzed using paired Wilcoxon signed-rank and Mann-Whitney U tests, respectively. The ability of lymphocyte subsets to predict treatment efficacy was analyzed using receiver operating characteristic curve and logistic regression.
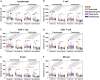
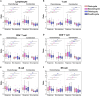
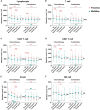
Results: The absolute counts of lymphocyte subsets in the response group significantly increased after the first cycle of chemotherapy or combination immunotherapy, whereas those in the non-response group showed persistent decreases. Ratios of lymphocyte subsets after the first treatment cycle to those at baseline were able to predict treatment efficacy early. Combination immunotherapy could increase lymphocyte counts compared to chemotherapy alone. In addition, patients with NSCLC receiving chemotherapy or combination immunotherapy for the first time mainly presented with elevated lymphocyte levels, whereas multiline patients showed continuous reductions.
Conclusion: Dynamic surveillance of lymphocyte subsets could reflect a more actual immune status and predict efficacy early. Combination immunotherapy protected lymphocyte levels from rapid decrease and patients undergoing multiline treatments were more prone to lymphopenia than those receiving first-line treatment. This study provides a reference for the early prediction of the efficacy of clinical tumor treatment for timely combination of immunotherapy or the improvement of immune status.
Keywords: NSCLC; chemotherapy; efficacy prediction; immunotherapy; lymphocyte subsets.
Copyright © 2024 Zhen, Wang, Qin, Lu, Yang and Zhang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The reviewer LZ declared a shared parent affiliation with the authors, and the reviewer XR declared a past co-authorship with the author YZ at the time of the review. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures

References
-
- Brahmer JR, Lee JS, Ciuleanu TE, Bernabe Caro R, Nishio M, Urban L, et al. Five-year survival outcomes with nivolumab plus ipilimumab versus chemotherapy as first-line treatment for metastatic non-small-cell lung cancer in CheckMate 227. J Clin Oncol. (2023) 41:1200–12. doi: 10.1200/JCO.22.01503 - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

