The relationship of ALPK1, hyaluronic acid and M1 macrophage polarization in the temporomandibular joint synovitis
- PMID: 38494837
- PMCID: PMC10945073
- DOI: 10.1111/jcmm.18172
The relationship of ALPK1, hyaluronic acid and M1 macrophage polarization in the temporomandibular joint synovitis
Abstract
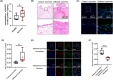
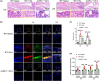
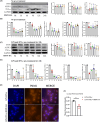
M1 macrophage polarization and synovitis play an important role in the pathogenesis of temporomandibular joint osteoarthritis (TMJOA). Reduced molecular weight of hyaluronic acid (HA) in synovial fluid of patients with TMJOA. In addition, high molecular weight hyaluronic acid (HMW-HA) is often used clinically to treat TMJ inflammation. As a pattern recognition receptor of the cytoplasm, ALPK1 was found to be pro-inflammatory in a variety of diseases. However, the relationship of ALPK1, HA and M1 macrophage polarization in TMJ synovitis remains unclear. We aimed to investigate the role of ALPK1 and HA in macrophage polarization and TMJ synovitis and the underlying mechanisms. The results demonstrated that ALPK1 was highly upregulated in the synovial macrophages in the inflamed TMJ synovium of patients. Low molecular weight hyaluronic acid (LMW-HA) promoted the expression of ALPK1 and M1 macrophage-associated genes. Besides, rhALPK1 promoted the expression of M1 macrophage-associated factors and the nuclear translocation of PKM2. Furthermore, ALPK1 knockout mice exhibited limited infiltration of macrophages and decreased expression levels of M1 macrophage-associated genes in CFA-induced TMJ synovitis. While HMW-HA inhibited the expression of ALPK1 and M1 macrophage polarization. Our results elucidated that ALPK1 promoted TMJ synovitis by promoting nuclear PKM2-mediated M1 macrophage polarization, whereas HMW-HA inhibited the expression of ALPK1 as well as M1 macrophage polarization.
Keywords: ALPK1; PKM2; hyaluronic acid; macrophage polarization; temporomandibular joint synovitis.
© 2024 The Authors. Journal of Cellular and Molecular Medicine published by Foundation for Cellular and Molecular Medicine and John Wiley & Sons Ltd.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Figures




Similar articles
-
High-Molecular-Weight Hyaluronic Acid Inhibits IL-1β-Induced Synovial Inflammation and Macrophage Polarization through the GRP78-NF-κB Signaling Pathway.Int J Mol Sci. 2021 Nov 3;22(21):11917. doi: 10.3390/ijms222111917. Int J Mol Sci. 2021. PMID: 34769349 Free PMC article.
-
Estradiol promotes M1-like macrophage activation through cadherin-11 to aggravate temporomandibular joint inflammation in rats.J Immunol. 2015 Mar 15;194(6):2810-8. doi: 10.4049/jimmunol.1303188. Epub 2015 Feb 13. J Immunol. 2015. PMID: 25681337
-
IL-38 suppresses macrophage M1 polarization to ameliorate synovial inflammation in the TMJ via GLUT-1 inhibition.Int Immunopharmacol. 2023 Sep;122:110619. doi: 10.1016/j.intimp.2023.110619. Epub 2023 Jul 17. Int Immunopharmacol. 2023. PMID: 37463548
-
Inflammation and Temporomandibular Joint Derangement.Biol Pharm Bull. 2019;42(4):538-542. doi: 10.1248/bpb.b18-00442. Biol Pharm Bull. 2019. PMID: 30930413 Review.
-
Mechanisms of Action and Efficacy of Hyaluronic Acid, Corticosteroids and Platelet-Rich Plasma in the Treatment of Temporomandibular Joint Osteoarthritis-A Systematic Review.Int J Mol Sci. 2021 Jul 9;22(14):7405. doi: 10.3390/ijms22147405. Int J Mol Sci. 2021. PMID: 34299024 Free PMC article.
Cited by
-
The importance of ALPK1 kinase functionality as a potential biomarker for inflammatory diseases.Mol Biol Rep. 2025 Jun 10;52(1):575. doi: 10.1007/s11033-025-10528-w. Mol Biol Rep. 2025. PMID: 40493324 Review.
-
Stem Cells from Human Exfoliated Deciduous Teeth-Derived Exosomes for the Treatment of Acute Liver Injury and Liver Fibrosis.ACS Appl Mater Interfaces. 2025 Mar 26;17(12):17948-17964. doi: 10.1021/acsami.4c19748. Epub 2025 Mar 14. ACS Appl Mater Interfaces. 2025. PMID: 40087139 Free PMC article.
-
S-propargyl-cysteine attenuates temporomandibular joint osteoarthritis by regulating macrophage polarization via Inhibition of JAK/STAT signaling.Mol Med. 2025 Apr 7;31(1):128. doi: 10.1186/s10020-025-01186-6. Mol Med. 2025. PMID: 40197110 Free PMC article.
-
DPSCs modulate synovial macrophage polarization and efferocytosis via PINK1/Parkin-dependent mitophagy.Stem Cell Res Ther. 2025 Jul 9;16(1):356. doi: 10.1186/s13287-025-04468-2. Stem Cell Res Ther. 2025. PMID: 40629418 Free PMC article.
-
Targeting Macrophage Polarization for Reinstating Homeostasis following Tissue Damage.Int J Mol Sci. 2024 Jul 2;25(13):7278. doi: 10.3390/ijms25137278. Int J Mol Sci. 2024. PMID: 39000385 Free PMC article. Review.
References
-
- Berenbaum F. Osteoarthritis as an inflammatory disease (osteoarthritis is not osteoarthrosis!). Osteoarthr Cartil. 2013;21:16‐21. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

