Co-Delivery of siRNA and Docetaxel to Cancer Cells by NLC for Therapy
- PMID: 38496987
- PMCID: PMC10938417
- DOI: 10.1021/acsomega.3c09098
Co-Delivery of siRNA and Docetaxel to Cancer Cells by NLC for Therapy
Abstract
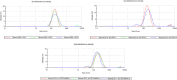
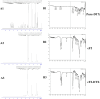
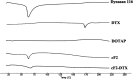
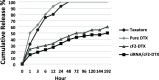
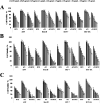
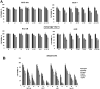
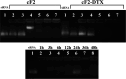
The present study aims to develop a delivery system that can carry small interference RNA (siRNA) with small-molecule chemotherapeutic drugs, which can be used in cancer treatment. The drug delivery system combines the advantages of a therapeutic agent with two different mechanisms to ensure that it is used efficiently for cancer therapy. In this study, a nanostructured lipid carrier system was prepared, Docetaxel was loaded to these systems, and the Eph siRNA was adsorbed to the outer surface. In addition, DOTAP was added to the lipophilic phase to load a positive charge on the lipidic structure for interaction with the cells. Moreover, characterization, cytotoxicity, and transfection procedures were performed on the whole system. This candidate system was also compared to Taxotere, which is the first approved Docetaxel-containing drug on the market. Given the results, it was determined that the particle size of NLC-DTX was 165.3 ± 3.5 nm, the ζ potential value was 38.2 ± 1.7 mV, and the PDI was 0.187 ± 0.024. Entrapment efficacy of nanoparticles was found to be 92.89 ± 0.21%. It was determined that the lipidic system prepared in vitro release analyses were able to provide sustained release and exhibit cytotoxicity, even at doses lower than the dose used for Taxotere. The formulations prepared had a higher level of effect on cells when compared with pure DTX and Taxotere, but they also exhibited time-dependent cytotoxicity. It was also observed that the use of Eph siRNA together with the chemotherapeutic agent via formulation also contributed to this cell death. The results of the present study indicate that there is a promising carrier system in order to deliver hydrophilic nucleic acids, such as siRNA, together with lipophilic drugs in cancer treatment.
© 2024 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

Similar articles
-
Glioblastoma multiforme targeted delivery of docetaxel using bevacizumab-modified nanostructured lipid carriers impair in vitro cell growth and in vivo tumor progression.Int J Pharm. 2022 Apr 25;618:121682. doi: 10.1016/j.ijpharm.2022.121682. Epub 2022 Mar 17. Int J Pharm. 2022. PMID: 35307470
-
Redox-responsive polymeric micelles formed by conjugating gambogic acid with bioreducible poly(amido amine)s for the co-delivery of docetaxel and MMP-9 shRNA.Acta Biomater. 2018 Mar 1;68:137-153. doi: 10.1016/j.actbio.2017.12.028. Epub 2017 Dec 26. Acta Biomater. 2018. PMID: 29288085
-
Nanostructured lipid carriers as novel carrier for parenteral delivery of docetaxel.Colloids Surf B Biointerfaces. 2011 Jul 1;85(2):262-9. doi: 10.1016/j.colsurfb.2011.02.038. Epub 2011 Mar 8. Colloids Surf B Biointerfaces. 2011. PMID: 21435845
-
Pharmacokinetics and in vitro/in vivo antitumor efficacy of aptamer-targeted Ecoflex® nanoparticles for docetaxel delivery in ovarian cancer.Int J Nanomedicine. 2018 Jan 23;13:493-504. doi: 10.2147/IJN.S152474. eCollection 2018. Int J Nanomedicine. 2018. PMID: 29416331 Free PMC article.
-
Formulation and in vitro characterization of domperidone loaded solid lipid nanoparticles and nanostructured lipid carriers.Daru. 2011;19(1):23-32. Daru. 2011. PMID: 22615636 Free PMC article.
Cited by
-
Leveraging nanostructured lipid carriers to enhance targeted delivery and efficacy in breast cancer therapy: a comprehensive review.Naunyn Schmiedebergs Arch Pharmacol. 2025 Jan;398(1):449-468. doi: 10.1007/s00210-024-03408-w. Epub 2024 Aug 28. Naunyn Schmiedebergs Arch Pharmacol. 2025. PMID: 39196394 Review.
References
-
- Erdemoğlu N.; Şener B. The antiumor effects of the taxane class compounds. J. Fac. Pharm. Ankara Univ. 2000, 29 (1), 77–90. 10.1501/Eczfak_0000000343. - DOI
-
- Nikolskaya E.-D.; Zhunina O.-A.; Yabbarov N.-G.; tereshchenko O.-G.; Godovanny A.-V.; Gukasova N.-V.; Severin E.-S. The docetaxel polymeric form and its antitumor activity. Russ. J. Bioorg. Chem. 2017, 43, 278–285. 10.1134/S1068162017030116. - DOI
LinkOut - more resources
Full Text Sources
Miscellaneous
