Echinocandin persistence directly impacts the evolution of resistance and survival of the pathogenic fungus Candida glabrata
- PMID: 38501869
- PMCID: PMC11005346
- DOI: 10.1128/mbio.00072-24
Echinocandin persistence directly impacts the evolution of resistance and survival of the pathogenic fungus Candida glabrata
Abstract
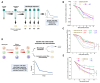
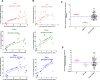
Recent epidemiological studies documented an alarming increase in the prevalence of echinocandin-resistant (ECR) Candida glabrata blood isolates. ECR isolates are known to arise from a minor subpopulation of a clonal population, termed echinocandin persisters. Although it is believed that isolates with a higher echinocandin persistence (ECP) are more likely to develop ECR, the implication of ECP needs to be better understood. Moreover, replacing laborious and time-consuming traditional approaches to determine ECP levels with rapid, convenient, and reliable tools is imperative to advance our understanding of this emerging concept in clinical practice. Herein, using extensive ex vivo and in vivo systemic infection models, we showed that high ECP isolates are less effectively cleared by micafungin treatment and exclusively give rise to ECR colonies. Additionally, we developed a flow cytometry-based tool that takes advantage of a SYTOX-based assay for the stratification of ECP levels. Once challenged with various collections of echinocandin-susceptible blood isolates, our assay reliably differentiated ECP levels in vitro and predicted ECP levels in real time under ex vivo and in vivo conditions when compared to traditional methods relying on colony-forming unit counting. Given the high and low ECP predictive values of 92.3% and 82.3%, respectively, our assay showed a high agreement with traditional approach. Collectively, our study supports the concept of ECP level determination in clinical settings and provides a robust tool scalable for high-throughput settings. Application of this tool facilitates the interrogation of mutant and drug libraries to further our understanding of persister biology and designing anti-persister therapeutics.
Importance: Candida glabrata is a prevalent fungal pathogen able to replicate inside macrophages and rapidly develop resistance against frontline antifungal echinocandins. Multiple studies have shown that echinocandin resistance is fueled by the survival of a small subpopulation of susceptible cells surviving lethal concentrations of echinocandins. Importantly, bacterial pathogens that exhibit high antibiotic persistence also impose a high burden and generate more antibiotic-resistant colonies. Nonetheless, the implications of echinocandin persistence (ECP) among the clinical isolates of C. glabrata have not been defined. Additionally, ECP level determination relies on a laborious and time-consuming method, which is prone to high variation. By exploiting in vivo systemic infection and ex vivo models, we showed that C. glabrata isolates with a higher ECP are associated with a higher burden and more likely develop echinocandin resistance upon micafungin treatment. Additionally, we developed an assay that reliably determines ECP levels in real time. Therefore, our study identified C. glabrata isolates displaying high ECP levels as important entities and provided a reliable and convenient tool for measuring echinocandin persistence, which is extendable to other fungal and bacterial pathogens.
Keywords: Candida glabrata; echinocandin; ex vivo; in vivo; persistence; tolerance.
Conflict of interest statement
“Michael K. Mansour consults for Clear Creek Bio, Day Zero Diagnostics, Safi, GenMark Diagnostics, NED Biosystems, and Vericel. He received grants from Karius, Danaher, Genentech, and Thermo-Fisher Scientific. He is a medical writer for UpToDate. The remaining authors have nothing to report.”
Figures



References
-
- Seider K, Brunke S, Schild L, Jablonowski N, Wilson D, Majer O, Barz D, Haas A, Kuchler K, Schaller M, Hube B. 2011. The facultative intracellular pathogen Candida glabrata subverts macrophage cytokine production and phagolysosome maturation. J Immunol 187:3072–3086. doi:10.4049/jimmunol.1003730 - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

