Antiemetic activity of abietic acid possibly through the 5HT3 and muscarinic receptors interaction pathways
- PMID: 38503897
- PMCID: PMC10951218
- DOI: 10.1038/s41598-024-57173-0
Antiemetic activity of abietic acid possibly through the 5HT3 and muscarinic receptors interaction pathways
Abstract
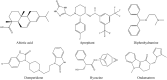
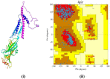
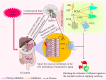
The present study was designed to evaluate the antiemetic activity of abietic acid (AA) using in vivo and in silico studies. To assess the effect, doses of 50 mg/kg b.w. copper sulfate (CuSO4⋅5H2O) were given orally to 2-day-old chicks. The test compound (AA) was given orally at two doses of 20 and 40 mg/kg b.w. On the other hand, aprepitant (16 mg/kg), domperidone (6 mg/kg), diphenhydramine (10 mg/kg), hyoscine (21 mg/kg), and ondansetron (5 mg/kg) were administered orally as positive controls (PCs). The vehicle was used as a control group. Combination therapies with the referral drugs were also given to three separate groups of animals to see the synergistic and antagonizing activity of the test compound. Molecular docking and visualization of ligand-receptor interaction were performed using different computational tools against various emesis-inducing receptors (D2, D3, 5HT3, H1, and M1-M5). Furthermore, the pharmacokinetics and toxicity properties of the selected ligands were predicted by using the SwissADME and Protox-II online servers. Findings indicated that AA dose-dependently enhances the latency of emetic retching and reduces the number of retching compared to the vehicle group. Among the different treatments, animals treated with AA (40 mg/kg) exhibited the highest latency (98 ± 2.44 s) and reduced the number of retching (11.66 ± 2.52 times) compared to the control groups. Additionally, the molecular docking study indicated that AA exhibits the highest binding affinity (- 10.2 kcal/mol) toward the M4 receptors and an elevated binding affinity toward the receptors 5HT3 (- 8.1 kcal/mol), M1 (- 7.7 kcal/mol), M2 (- 8.7 kcal/mol), and H1 (- 8.5 kcal/mol) than the referral ligands. Taken together, our study suggests that AA has potent antiemetic effects by interacting with the 5TH3 and muscarinic receptor interaction pathways. However, additional extensive pre-clinical and clinical studies are required to evaluate the efficacy and toxicity of AA.
Keywords: Gallus gallus domesticus; Abietic acid; Emesis; Molecular docking; Vomiting.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures





Similar articles
-
Synergistic Antiemetic Effects of Nerolidol on Domperidone, Hyoscine, and Ondansetron: In Vivo and in Silico Investigations on Receptor Binding Affinity.ChemistryOpen. 2025 Mar;14(3):e202400345. doi: 10.1002/open.202400345. Epub 2024 Nov 12. ChemistryOpen. 2025. PMID: 39533458 Free PMC article.
-
Exploring the Antiemetic Potential of Caffeic Acid: A Combined In Vivo and Computational Approach.Neurogastroenterol Motil. 2025 Jun;37(6):e70003. doi: 10.1111/nmo.70003. Epub 2025 Feb 17. Neurogastroenterol Motil. 2025. PMID: 39962751
-
Assessment of antiemetic activity of dihydrocoumarin: In vivo and in silico approaches on receptor binding affinity and modulatory effects.J Pharmacol Toxicol Methods. 2024 Nov-Dec;130:107561. doi: 10.1016/j.vascn.2024.107561. Epub 2024 Sep 24. J Pharmacol Toxicol Methods. 2024. PMID: 39326519
-
Progress in reducing nausea and emesis. Comparisons of ondansetron (Zofran), granisetron (Kytril), and tropisetron (Navoban).Cancer. 1995 Aug 1;76(3):343-57. doi: 10.1002/1097-0142(19950801)76:3<343::aid-cncr2820760302>3.0.co;2-v. Cancer. 1995. PMID: 8625113 Review.
-
[Recent improvements in antiemetic therapy].Tumori. 1997;83(2 Suppl):S3-14. Tumori. 1997. PMID: 9235727 Review. Italian.
Cited by
-
Anxiolytic Efficacy of Indirubin: In Vivo Approach Along with Receptor Binding Profiling and Molecular Interaction with GABAergic Pathways.ChemistryOpen. 2025 Feb;14(2):e202400290. doi: 10.1002/open.202400290. Epub 2024 Oct 25. ChemistryOpen. 2025. PMID: 39460441 Free PMC article.
-
Synergistic Sedative Activity of Indirubin on Diazepam in Thiopental Sodium-induced Sleeping Mice: A Potential GABAergic Transmission.Mol Neurobiol. 2025 Jul 1. doi: 10.1007/s12035-025-05176-x. Online ahead of print. Mol Neurobiol. 2025. PMID: 40593261
-
Tangeretin Improves the Memory of Swiss Mice, Suggesting Potential Molecular Interventions Through Animal Behavior Assessments and In Silico Studies.Brain Behav. 2025 May;15(5):e70516. doi: 10.1002/brb3.70516. Brain Behav. 2025. PMID: 40320970 Free PMC article.
-
Sedative Effects of Daidzin, Possibly Through the GABAA Receptor Interaction Pathway: In Vivo Approach with Molecular Dynamic Simulations.J Mol Neurosci. 2024 Sep 4;74(3):83. doi: 10.1007/s12031-024-02261-z. J Mol Neurosci. 2024. PMID: 39230641
-
Caffeine and sclareol take the edge off the sedative effects of linalool, possibly through the GABAA interaction pathway: molecular insights through in vivo and in silico studies.Naunyn Schmiedebergs Arch Pharmacol. 2025 Aug;398(8):10997-11009. doi: 10.1007/s00210-025-03915-4. Epub 2025 Mar 11. Naunyn Schmiedebergs Arch Pharmacol. 2025. PMID: 40067442
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

