Effect of a self-developed fixation device on preventing endotracheal intubation-related pressure injury: a randomised controlled trial
- PMID: 38504251
- PMCID: PMC10953139
- DOI: 10.1186/s13054-024-04874-7
Effect of a self-developed fixation device on preventing endotracheal intubation-related pressure injury: a randomised controlled trial
Abstract
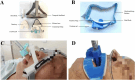
Objective: To evaluate the effects of our self-developed endotracheal tube fixation device in mechanically ventilated patients.
Methods: In a dual-centre randomised controlled trial, patients who were expected to require mechanical ventilation for over 48 h were assigned to the observation group (using self-developed device) or the control group (using the traditional device). The primary endpoint was the incidence of endotracheal intubation-related pressure injury (EIRPI).
Results: Fifty-one patients in the observation group and 54 patients in the control group were analysed. The incidence of EIRPI was 7.8% in the observation group and 33.3% in the control group (p = 0.001). Lip pressure injury (PI) occurred in 0 versus 14 (25.9%) patients in the observation versus control groups (p < 0.001). Both oral-mucosal and facial PIs were similar between the two groups.
Conclusions: The use of the novel device reduced the incidence of EIRPI, especially lip PI. Trial registration Chinese Clinical Trial Registry ChiCTR2300078132. Registered on 29 November 2023.
Keywords: EIRPI; Endotracheal tube; Fixation; Lip; Pressure injury.
© 2024. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
Similar articles
-
Risk Factors of Endotracheal Intubation-Related Pressure Injury among Patients Admitted to the ICU.Adv Skin Wound Care. 2021 Mar 1;34(3):144-148. doi: 10.1097/01.ASW.0000732896.29121.06. Adv Skin Wound Care. 2021. PMID: 33587475
-
The effect of adhesive tape versus endotracheal tube fastener in critically ill adults: the endotracheal tube securement (ETTS) randomized controlled trial.Crit Care. 2019 May 7;23(1):161. doi: 10.1186/s13054-019-2440-7. Crit Care. 2019. PMID: 31064406 Free PMC article. Clinical Trial.
-
Risk factors for oral mucosal pressure injury associated with endotracheal tubes in intensive care unit patients: A single-centre longitudinal study with brief follow-up.Nurs Crit Care. 2025 Mar;30(2):e70009. doi: 10.1111/nicc.70009. Nurs Crit Care. 2025. PMID: 40068960
-
Prevention of Endotracheal Tube-Related Pressure Injury: A Systematic Review and Meta-analysis.Am J Crit Care. 2022 Sep 1;31(5):416-424. doi: 10.4037/ajcc2022644. Am J Crit Care. 2022. PMID: 36045034
-
Nasal protection strategy reduces the incidence of nasal pressure injuries during nasotracheal intubation: Meta-analysis with trial sequential analysis.Medicine (Baltimore). 2022 Oct 7;101(40):e30638. doi: 10.1097/MD.0000000000030638. Medicine (Baltimore). 2022. PMID: 36221363 Free PMC article.
Cited by
-
Application of double-sleeve endotracheal tube in infection control for icu patients: a randomized controlled trial.Head Face Med. 2025 Feb 26;21(1):12. doi: 10.1186/s13005-025-00488-8. Head Face Med. 2025. PMID: 40011996 Free PMC article. Clinical Trial.
-
Innovative Tracheal Tube Design Enhances Oral Health in Intubated Patients.Int Wound J. 2025 Jul;22(7):e70701. doi: 10.1111/iwj.70701. Int Wound J. 2025. PMID: 40603266 Free PMC article. Clinical Trial.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

