Combined KRAS-MAPK pathway inhibitors and HER2-directed drug conjugate is efficacious in pancreatic cancer
- PMID: 38509064
- PMCID: PMC10954758
- DOI: 10.1038/s41467-024-46811-w
Combined KRAS-MAPK pathway inhibitors and HER2-directed drug conjugate is efficacious in pancreatic cancer
Abstract
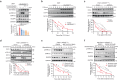
Targeting the mitogen-activated protein kinase (MAPK) cascade in pancreatic ductal adenocarcinoma (PDAC) remains clinically unsuccessful. We aim to develop a MAPK inhibitor-based therapeutic combination with strong preclinical efficacy. Utilizing a reverse-phase protein array, we observe rapid phospho-activation of human epidermal growth factor receptor 2 (HER2) in PDAC cells upon pharmacological MAPK inhibition. Mechanistically, MAPK inhibitors lead to swift proteasomal degradation of dual-specificity phosphatase 6 (DUSP6). The carboxy terminus of HER2, containing a TEY motif also present in extracellular signal-regulated kinase 1/2 (ERK1/2), facilitates binding with DUSP6, enhancing its phosphatase activity to dephosphorylate HER2. In the presence of MAPK inhibitors, DUSP6 dissociates from the protective effect of the RING E3 ligase tripartite motif containing 21, resulting in its degradation. In PDAC patient-derived xenograft (PDX) models, combining ERK and HER inhibitors slows tumour growth and requires cytotoxic chemotherapy to achieve tumour regression. Alternatively, MAPK inhibitors with trastuzumab deruxtecan, an anti-HER2 antibody conjugated with cytotoxic chemotherapy, lead to sustained tumour regression in most tested PDXs without causing noticeable toxicity. Additionally, KRAS inhibitors also activate HER2, supporting testing the combination of KRAS inhibitors and trastuzumab deruxtecan in PDAC. This study identifies a rational and promising therapeutic combination for clinical testing in PDAC patients.
© 2024. The Author(s).
Conflict of interest statement
A.W.-G. is currently on sabbatical from Washington University and employed by Jacobio Pharmaceuticals, Inc. Deborah Knoerzer was employed for BioMed Valley Discovery. The remaining authors declare no competing interests.
Figures









References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

