In vitro and in vivo studies on the effect of a mitochondrial fusion promoter on Leydig cell integrity and function
- PMID: 38511146
- PMCID: PMC10950900
- DOI: 10.3389/ftox.2024.1357857
In vitro and in vivo studies on the effect of a mitochondrial fusion promoter on Leydig cell integrity and function
Abstract
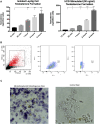
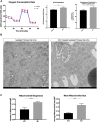
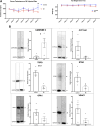
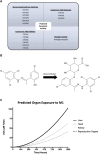
Background: The interstitial testicular Leydig cells are responsible for the production of testosterone, which functionally deteriorate with normal aging. Decreased expression of mitochondrial steroidogenic interactome proteins and diminished mitochondrial function in aging Leydig cells suggest that mitochondrial dynamics play a role in maintaining adequate levels of testosterone. Optic atrophy 1 (OPA1) protein regulates mitochondrial dynamics and cristae formation in many cell types. Previous studies showed that increasing OPA1 expression in dysfunctional Leydig cells restored mitochondrial function and recovered androgen production to levels found in healthy Leydig cells. These findings suggested that mitochondrial dynamics may be a promising target to ameliorate diminished testosterone levels in aging males. Methods: We used twelve-month-old rats to explore the relationship between mitochondrial dynamics and Leydig cell function. Isolated Leydig cells from aged rats were treated ex vivo with the cell-permeable mitochondrial fusion promoter 4-Chloro-2-(1-(2-(2,4,6-trichlorophenyl)hydrazono)ethyl) phenol (mitochondrial fusion promoter M1), which enhances mitochondrial tubular network formation. In parallel, rats were treated with 2 mg/kg/day M1 for 6 weeks before Leydig cells were isolated. Results: Ex vivo M1-treated cells showed enhanced mitochondrial tubular network formation by transmission electron microscopy, enhanced Leydig cell mitochondrial integrity, improved mitochondrial function, and higher testosterone biosynthesis compared to controls. However, in vivo treatment of aged rats with M1 not only failed to re-establish testosterone levels to that of young rats, it also led to further reduction of testosterone levels and increased apoptosis, suggesting M1 toxicity in the testis. The in vivo M1 toxicity seemed to be tissue-specific, however. Conclusion: Promoting mitochondrial fusion may be one approach to enhancing cell health and wellbeing with aging, but more investigations are warranted. Our findings suggest that fusion promoters could potentially enhance the productivity of aged Leydig cells when carefully regulated.
Keywords: aging; bioenergetics; hypogonadism; mitochondrial fusion; steroidogenesis; testosterone; toxicity.
Copyright © 2024 Garza, Sottas, Gukasyan and Papadopoulos.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Mitochondrial dynamics, Leydig cell function, and age-related testosterone deficiency.FASEB J. 2022 Dec;36(12):e22637. doi: 10.1096/fj.202201026R. FASEB J. 2022. PMID: 36349989
-
Aging-Related Increase of cGMP Disrupts Mitochondrial Homeostasis in Leydig Cells.J Gerontol A Biol Sci Med Sci. 2021 Jan 18;76(2):177-186. doi: 10.1093/gerona/glaa132. J Gerontol A Biol Sci Med Sci. 2021. PMID: 32459846
-
Triphenyl phosphate induced apoptosis of mice testicular Leydig cells and TM3 cells through ROS-mediated mitochondrial fusion inhibition.Ecotoxicol Environ Saf. 2023 May;256:114876. doi: 10.1016/j.ecoenv.2023.114876. Epub 2023 Apr 5. Ecotoxicol Environ Saf. 2023. PMID: 37027944
-
Regulation of Leydig cell steroidogenic function during aging.Biol Reprod. 2000 Oct;63(4):977-81. doi: 10.1095/biolreprod63.4.977. Biol Reprod. 2000. PMID: 10993816 Review.
-
Testosterone recovery therapy targeting dysfunctional Leydig cells.Andrology. 2023 Jul;11(5):816-825. doi: 10.1111/andr.13304. Epub 2022 Oct 10. Andrology. 2023. PMID: 36164998 Review.
Cited by
-
Age-related testosterone decline: mechanisms and intervention strategies.Reprod Biol Endocrinol. 2024 Nov 14;22(1):144. doi: 10.1186/s12958-024-01316-5. Reprod Biol Endocrinol. 2024. PMID: 39543598 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources

