Extracellular vesicles from vaginal Gardnerella vaginalis and Mobiluncus mulieris contain distinct proteomic cargo and induce inflammatory pathways
- PMID: 38514622
- PMCID: PMC10957959
- DOI: 10.1038/s41522-024-00502-y
Extracellular vesicles from vaginal Gardnerella vaginalis and Mobiluncus mulieris contain distinct proteomic cargo and induce inflammatory pathways
Abstract
Colonization of the vaginal space with bacteria such as Gardnerella vaginalis and Mobiluncus mulieris is associated with increased risk for STIs, bacterial vaginosis, and preterm birth, while Lactobacillus crispatus is associated with optimal reproductive health. Although host-microbe interactions are hypothesized to contribute to reproductive health and disease, the bacterial mediators that are critical to this response remain unclear. Bacterial extracellular vesicles (bEVs) are proposed to participate in host-microbe communication by providing protection of bacterial cargo, delivery to intracellular targets, and ultimately induction of immune responses from the host. We evaluated the proteome of bEVs produced in vitro from G. vaginalis, M. mulieris, and L. crispatus, identifying specific proteins of immunologic interest. We found that bEVs from each bacterial species internalize within cervical and vaginal epithelial cells, and that epithelial and immune cells express a multi-cytokine response when exposed to bEVs from G. vaginalis and M. mulieris but not L. crispatus. Further, we demonstrate that the inflammatory response induced by G. vaginalis and M. mulieris bEVs is TLR2-specific. Our results provide evidence that vaginal bacteria communicate with host cells through secreted bEVs, revealing a mechanism by which bacteria lead to adverse reproductive outcomes associated with inflammation. Elucidating host-microbe interactions in the cervicovaginal space will provide further insight into the mechanisms contributing to microbiome-mediated adverse outcomes and may reveal new therapeutic targets.
© 2024. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


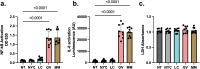
Similar articles
-
Intrauterine colonization with Gardnerella vaginalis and Mobiluncus mulieris induces maternal inflammation but not preterm birth in a mouse model.Am J Reprod Immunol. 2023 Aug;90(2):e13749. doi: 10.1111/aji.13749. Am J Reprod Immunol. 2023. PMID: 37491927 Free PMC article.
-
Gardnerella vaginalis alters cervicovaginal epithelial cell function through microbe-specific immune responses.Microbiome. 2022 Aug 4;10(1):119. doi: 10.1186/s40168-022-01317-9. Microbiome. 2022. PMID: 35922830 Free PMC article.
-
Mobiluncus mulieris alters the transcriptomic profile of cervicovaginal epithelial cells, shedding light on molecular drivers of adverse reproductive outcomes.NPJ Biofilms Microbiomes. 2025 Aug 8;11(1):154. doi: 10.1038/s41522-025-00784-w. NPJ Biofilms Microbiomes. 2025. PMID: 40775239 Free PMC article.
-
Influence of Biofilm Formation by Gardnerella vaginalis and Other Anaerobes on Bacterial Vaginosis.J Infect Dis. 2015 Dec 15;212(12):1856-61. doi: 10.1093/infdis/jiv338. Epub 2015 Jun 16. J Infect Dis. 2015. PMID: 26080369 Review.
-
Gardnerella vaginalis: characteristics, clinical considerations, and controversies.Clin Microbiol Rev. 1992 Jul;5(3):213-37. doi: 10.1128/CMR.5.3.213. Clin Microbiol Rev. 1992. PMID: 1498765 Free PMC article. Review.
Cited by
-
Harnessing microbial nanobiotics: Lactobacillus extracellular vesicles as next-generation therapeutics across physiological systems.World J Microbiol Biotechnol. 2025 Jul 11;41(7):261. doi: 10.1007/s11274-025-04481-w. World J Microbiol Biotechnol. 2025. PMID: 40640466 Review.
-
Bacterial extracellular vesicles as a tunable platform for vaginal drug delivery.bioRxiv [Preprint]. 2025 Jun 9:2025.06.09.658669. doi: 10.1101/2025.06.09.658669. bioRxiv. 2025. PMID: 40661620 Free PMC article. Preprint.
-
Vaginal bacteria-derived extracellular vesicles diffuse through human cervicovaginal mucus to enable bacterial signaling to upper female reproductive tract tissues.bioRxiv [Preprint]. 2025 May 1:2025.04.28.651063. doi: 10.1101/2025.04.28.651063. bioRxiv. 2025. PMID: 40654697 Free PMC article. Preprint.
-
The role of extracellular vesicles in the pathogenesis of gynecological cancer.Front Oncol. 2024 Sep 26;14:1477610. doi: 10.3389/fonc.2024.1477610. eCollection 2024. Front Oncol. 2024. PMID: 39391238 Free PMC article. Review.
-
Harnessing Microbiome, Bacterial Extracellular Vesicle, and Artificial Intelligence for Polycystic Ovary Syndrome Diagnosis and Management.Biomolecules. 2025 Jun 7;15(6):834. doi: 10.3390/biom15060834. Biomolecules. 2025. PMID: 40563474 Free PMC article. Review.
References
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

