Global, regional, and national age-specific progress towards the 2020 milestones of the WHO End TB Strategy: a systematic analysis for the Global Burden of Disease Study 2021
- PMID: 38518787
- PMCID: PMC11187709
- DOI: 10.1016/S1473-3099(24)00007-0
Global, regional, and national age-specific progress towards the 2020 milestones of the WHO End TB Strategy: a systematic analysis for the Global Burden of Disease Study 2021
Abstract
Background: Global evaluations of the progress towards the WHO End TB Strategy 2020 interim milestones on mortality (35% reduction) and incidence (20% reduction) have not been age specific. We aimed to assess global, regional, and national-level burdens of and trends in tuberculosis and its risk factors across five separate age groups, from 1990 to 2021, and to report on age-specific progress between 2015 and 2020.
Methods: We used the Global Burden of Diseases, Injuries, and Risk Factors Study 2021 (GBD 2021) analytical framework to compute age-specific tuberculosis mortality and incidence estimates for 204 countries and territories (1990-2021 inclusive). We quantified tuberculosis mortality among individuals without HIV co-infection using 22 603 site-years of vital registration data, 1718 site-years of verbal autopsy data, 825 site-years of sample-based vital registration data, 680 site-years of mortality surveillance data, and 9 site-years of minimally invasive tissue sample (MITS) diagnoses data as inputs into the Cause of Death Ensemble modelling platform. Age-specific HIV and tuberculosis deaths were established with a population attributable fraction approach. We analysed all available population-based data sources, including prevalence surveys, annual case notifications, tuberculin surveys, and tuberculosis mortality, in DisMod-MR 2.1 to produce internally consistent age-specific estimates of tuberculosis incidence, prevalence, and mortality. We also estimated age-specific tuberculosis mortality without HIV co-infection that is attributable to the independent and combined effects of three risk factors (smoking, alcohol use, and diabetes). As a secondary analysis, we examined the potential impact of the COVID-19 pandemic on tuberculosis mortality without HIV co-infection by comparing expected tuberculosis deaths, modelled with trends in tuberculosis deaths from 2015 to 2019 in vital registration data, with observed tuberculosis deaths in 2020 and 2021 for countries with available cause-specific mortality data.
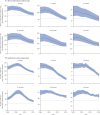
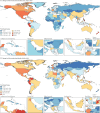
Findings: We estimated 9·40 million (95% uncertainty interval [UI] 8·36 to 10·5) tuberculosis incident cases and 1·35 million (1·23 to 1·52) deaths due to tuberculosis in 2021. At the global level, the all-age tuberculosis incidence rate declined by 6·26% (5·27 to 7·25) between 2015 and 2020 (the WHO End TB strategy evaluation period). 15 of 204 countries achieved a 20% decrease in all-age tuberculosis incidence between 2015 and 2020, eight of which were in western sub-Saharan Africa. When stratified by age, global tuberculosis incidence rates decreased by 16·5% (14·8 to 18·4) in children younger than 5 years, 16·2% (14·2 to 17·9) in those aged 5-14 years, 6·29% (5·05 to 7·70) in those aged 15-49 years, 5·72% (4·02 to 7·39) in those aged 50-69 years, and 8·48% (6·74 to 10·4) in those aged 70 years and older, from 2015 to 2020. Global tuberculosis deaths decreased by 11·9% (5·77 to 17·0) from 2015 to 2020. 17 countries attained a 35% reduction in deaths due to tuberculosis between 2015 and 2020, most of which were in eastern Europe (six countries) and central Europe (four countries). There was variable progress by age: a 35·3% (26·7 to 41·7) decrease in tuberculosis deaths in children younger than 5 years, a 29·5% (25·5 to 34·1) decrease in those aged 5-14 years, a 15·2% (10·0 to 20·2) decrease in those aged 15-49 years, a 7·97% (0·472 to 14·1) decrease in those aged 50-69 years, and a 3·29% (-5·56 to 9·07) decrease in those aged 70 years and older. Removing the combined effects of the three attributable risk factors would have reduced the number of all-age tuberculosis deaths from 1·39 million (1·28 to 1·54) to 1·00 million (0·703 to 1·23) in 2020, representing a 36·5% (21·5 to 54·8) reduction in tuberculosis deaths compared to those observed in 2015. 41 countries were included in our analysis of the impact of the COVID-19 pandemic on tuberculosis deaths without HIV co-infection in 2020, and 20 countries were included in the analysis for 2021. In 2020, 50 900 (95% CI 49 700 to 52 400) deaths were expected across all ages, compared to an observed 45 500 deaths, corresponding to 5340 (4070 to 6920) fewer deaths; in 2021, 39 600 (38 300 to 41 100) deaths were expected across all ages compared to an observed 39 000 deaths, corresponding to 657 (-713 to 2180) fewer deaths.
Interpretation: Despite accelerated progress in reducing the global burden of tuberculosis in the past decade, the world did not attain the first interim milestones of the WHO End TB Strategy in 2020. The pace of decline has been unequal with respect to age, with older adults (ie, those aged >50 years) having the slowest progress. As countries refine their national tuberculosis programmes and recalibrate for achieving the 2035 targets, they could consider learning from the strategies of countries that achieved the 2020 milestones, as well as consider targeted interventions to improve outcomes in older age groups.
Funding: Bill & Melinda Gates Foundation.
Copyright © 2024 The Author(s). Published by Elsevier Ltd. This is an Open Access article under the CC BY 4.0 license. Published by Elsevier Ltd.. All rights reserved.
Conflict of interest statement
Declaration of interests J R Andrews reports grants or contracts from the US National Institutes of Health. Z Basharat reports other financial interests as an employee of Alpha Genomics, outside the submitted work. P J G Bettencourt reports other financial or non-financial interests as a project reviewer at Botnar Foundation, outside the submitted work. L Cegolon reports grants or contracts from ORCHESTRA project WP5 (grant agreement number 101016167); payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing, or educational events from the University of Trieste (Italy) and University Pavia (Italy) academic lectures; and support for attending meetings or travel from the National Congress of the Italian Society of Occupational Medicine (Genoa: September, 2022; Turin: September, 2023) and the National Congress of the Italian Society of Public Health Medicine (Rome: May, 2023), all outside the submitted work. I Ilic reports support for this study from the Ministry of Education, Science and Technological development, Serbia (project number 175042, 2011–23). M Ilic reports support for this study from the Ministry of Education, Science and Technological development, Serbia (project number 451-03-47/2023-01/200111). N E Ismail reports unpaid leadership or fiduciary roles in board, society, committee, or advocacy groups, with the Malaysian Academy of Pharmacy as the Bursary and Council Member, outside the submitted work. K Krishan reports non-financial support from the UGC Centre of Advanced Study, CAS II, awarded to the Department of Anthropology, Panjab University (Chandigarh, India), outside the submitted work. M Lee reports support for this study from the Ministry of Education of South Korea and the National Research Foundation of Korea (NRF-2021R1I1A4A01057428) and Bio-convergence Technology Education Program through the Korea Institute for Advancement Technology (KIAT) funded by the Ministry of Trade, Industry and Energy (number P0017805). R J Maude reports support for this study from the Wellcome Trust, (grant number 220211) as it provides core funding for Mahidol Oxford Tropical Medicine Research Unit and contributes to their salary. L Monasta reports support for this study from the Italian Ministry of Health (Ricerca Corrente 34/2017), and payments made to the Institute for Maternal and Child Health IRCCS Burlo Garofolo. S Nomura reports support for this study from the Ministry of Education, Culture, Sports, Science and Technology of Japan (grant 21H03203) and from Precursory Research for Embryonic Science and Technology from the Japan Science and Technology Agency (grant JPMJPR22R8). V C F Pepito reports grants or contracts from the International Initiative for Impact Evaluation (3ie) and Sanofi Consumer Healthcare. L F Reyes reports grants or contracts, consulting fees, and payments or honoraria from MSD, GSK, and Pfizer, and support for attending meetings from GSK, outside the submitted work. O Rezahosseini reports grants or contracts from Rigshospitalet and A.P.Møllers fonden. J Ross reports grants or contracts from the US National Institutes of Health, the US Department of Veterans Affairs, and Merck. J P Silva reports salary support for this study from the Portuguese Foundation for Science and Technology. J A Singh reports consulting fees from AstraZeneca, Crealta/Horizon, Medisys, Fidia, PK Med, Two labs, Adept Field Solutions, Clinical Care Options, ClearView Healthcare Partners, Putnam Associates, Focus Forward, Guidehouse consulting (formerly Navigant), Spherix, MedIQ, Jupiter Life Science, UBM, Trio Health, Medscape, WebMD, Practice Point Communications, and the US National Institutes of Health and the American College of Rheumatology; payment of honoraria for lectures, presentations, speakers bureaus, manuscript writing, or education events as a member of the speaker's bureau of Simply Speaking; support for attending meetings as a past steering committee member of OMERACT (an international organisation that develops measures for clinical trials and receives funding from 12 pharmaceutical companies); participation on a data safety monitoring board or advisory board with the US Food and Drug Administration Arthritis Advisory Committee; leadership or fiduciary role in other board, society, committee, or advocacy groups, paid as a past steering committee member of OMERACT, unpaid as a Co-Chair of the Veterans Affairs Rheumatology Field Advisory Committee, and unpaid as an editor and Director of the UAB Cochrane Musculoskeletal Group Satellite Center on Network Meta-analysis; stock or stock options in atai Life Sciences, Kintara Therapeutics, Intelligent Biosolutions, Acumen Pharmaceuticals, TPT Global Tech, Vaxart Pharmaceuticals, Aytu BioPharma, Adaptimmune Therapeutics, GeoVax Labs, Pieris Pharmaceuticals, Enzolytics, Seres Therapeutics, Tonix Pharmaceuticals Holding, and Charlotte's Web Holdings; and previous stock options in Amarin, Viking, and Moderna Pharmaceuticals, all outside the submitted work. L K Stafford reports support for this study from the Bill & Melinda Gates Foundation and the Institute for Health Metrics and Evaluation (IHME). Z Wang reports grants or contracts from the Fred Hollows Foundation, Fonds de recherche du Québec - Santé, China Scholarship Council, and McGill University Global Health Scholars Program; consulting fees from the Fred Hollows Foundation; support for attending meetings or travel from McGill University (Graduate Mobility Award) and the Fred Hollows Foundation; and a leadership or fiduciary role in other board, society, committee, or advocacy groups, paid or unpaid as a member of the trainee advisory committee with the Consortium of Universities for Global Health, outside the submitted work. H Zhang reports grants or contracts from WHO research grants. M Zielińska reports other financial interests as an AstraZeneca employee, outside the submitted work.
Figures

References
-
- Uplekar M, Weil D, Lonnroth K, et al. WHO's new End TB Strategy. Lancet. 2015;385:1799–1801. - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

