Thiophene-Based Ligands for Specific Assignment of Distinct Aβ Pathologies in Alzheimer's Disease
- PMID: 38523263
- PMCID: PMC10995944
- DOI: 10.1021/acschemneuro.4c00021
Thiophene-Based Ligands for Specific Assignment of Distinct Aβ Pathologies in Alzheimer's Disease
Abstract
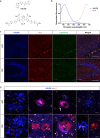
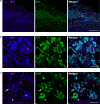
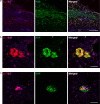
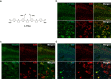
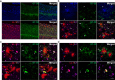
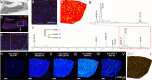
Aggregated species of amyloid-β (Aβ) are one of the pathological hallmarks in Alzheimer's disease (AD), and ligands that selectively target different Aβ deposits are of great interest. In this study, fluorescent thiophene-based ligands have been used to illustrate the features of different types of Aβ deposits found in AD brain tissue. A dual-staining protocol based on two ligands, HS-276 and LL-1, with different photophysical and binding properties, was developed and applied on brain tissue sections from patients affected by sporadic AD or familial AD associated with the PSEN1 A431E mutation. When binding to Aβ deposits, the ligands could easily be distinguished for their different fluorescence, and distinct staining patterns were revealed for these two types of AD. In sporadic AD, HS-276 consistently labeled all immunopositive Aβ plaques, whereas LL-1 mainly stained cored and neuritic Aβ deposits. In the PSEN1 A431E cases, each ligand was binding to specific types of Aβ plaques. The ligand-labeled Aβ deposits were localized in distinct cortical layers, and a laminar staining pattern could be seen. Biochemical characterization of the Aβ aggregates in the individual layers also showed that the variation of ligand binding properties was associated with certain Aβ peptide signatures. For the PSEN1 A431E cases, it was concluded that LL-1 was binding to cotton wool plaques, whereas HS-276 mainly stained diffuse Aβ deposits. Overall, our findings showed that a combination of ligands was essential to identify distinct aggregated Aβ species associated with different forms of AD.
Keywords: Alzheimer’s disease; amyloid-β; fluorescence; imaging mass spectrometry; ligands; protein aggregates.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


References
-
- Mann D. M. A.; Iwatsubo T.; Ihara Y.; Cairns N. J.; Lantos P. L.; Bogdanovic N.; et al. Predominant deposition of amyloid-β42(43) in plaques in cases of Alzheimer’s disease and hereditary cerebral hemorrhage associated with mutations in the amyloid precursor protein gene. Am. J. Pathol. 1996, 148, 1257–1266. - PMC - PubMed

