Cognitive decline, Aβ pathology, and blood-brain barrier function in aged 5xFAD mice
- PMID: 38532486
- PMCID: PMC10967049
- DOI: 10.1186/s12987-024-00531-x
Cognitive decline, Aβ pathology, and blood-brain barrier function in aged 5xFAD mice
Abstract
Background: Patients with Alzheimer's disease (AD) develop blood-brain barrier dysfunction to varying degrees. How aging impacts Aβ pathology, blood-brain barrier function, and cognitive decline in AD remains largely unknown. In this study, we used 5xFAD mice to investigate changes in Aβ levels, barrier function, and cognitive decline over time.
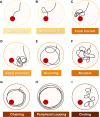
Methods: 5xFAD and wild-type (WT) mice were aged between 9.5 and 15.5 months and tested for spatial learning and reference memory with the Morris Water Maze (MWM). After behavior testing, mice were implanted with acute cranial windows and intravenously injected with fluorescent-labeled dextrans to assess their in vivo distribution in the brain by two-photon microscopy. Images were processed and segmented to obtain intravascular intensity, extravascular intensity, and vessel diameters as a measure of barrier integrity. Mice were sacrificed after in vivo imaging to isolate brain and plasma for measuring Aβ levels. The effect of age and genotype were evaluated for each assay using generalized or cumulative-linked logistic mixed-level modeling and model selection by Akaike Information Criterion (AICc). Pairwise comparisons were used to identify outcome differences between the two groups.
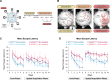
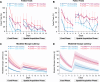
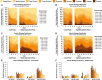
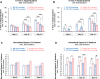
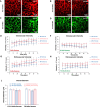
Results: 5xFAD mice displayed spatial memory deficits compared to age-matched WT mice in the MWM assay, which worsened with age. Memory impairment was evident in 5xFAD mice by 2-threefold higher escape latencies, twofold greater cumulative distances until they reach the platform, and twice as frequent use of repetitive search strategies in the pool when compared with age-matched WT mice. Presence of the rd1 allele worsened MWM performance in 5xFAD mice at all ages but did not alter the rate of learning or probe trial outcomes. 9.5-month-old 15.5-month-old 5xFAD mice had twofold higher brain Aβ40 and Aβ42 levels (p < 0.001) and 2.5-fold higher (p = 0.007) plasma Aβ40 levels compared to 9.5-month-old 5xFAD mice. Image analysis showed that vessel diameters and intra- and extravascular dextran intensities were not significantly different in 9.5- and 15.5-month-old 5xFAD mice compared to age-matched WT mice.
Conclusion: 5xFAD mice continue to develop spatial memory deficits and increased Aβ brain levels while aging. Given in vivo MP imaging limitations, further investigation with smaller molecular weight markers combined with advanced imaging techniques would be needed to reliably assess subtle differences in barrier integrity in aged mice.
Keywords: Pde6b rd1; 5xFAD; Aβ; Blood–Brain Barrier.
© 2024. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


Similar articles
-
Plasma S100β is a predictor for pathology and cognitive decline in Alzheimer's disease.Fluids Barriers CNS. 2025 Jan 9;22(1):4. doi: 10.1186/s12987-024-00615-8. Fluids Barriers CNS. 2025. PMID: 39789614 Free PMC article.
-
A longitudinal study of the 5xFAD mouse retina delineates Amyloid beta (Aβ)-mediated retinal pathology from age-related changes.Alzheimers Res Ther. 2025 Jun 19;17(1):136. doi: 10.1186/s13195-025-01784-w. Alzheimers Res Ther. 2025. PMID: 40537856 Free PMC article.
-
CSF tau and the CSF tau/ABeta ratio for the diagnosis of Alzheimer's disease dementia and other dementias in people with mild cognitive impairment (MCI).Cochrane Database Syst Rev. 2017 Mar 22;3(3):CD010803. doi: 10.1002/14651858.CD010803.pub2. Cochrane Database Syst Rev. 2017. PMID: 28328043 Free PMC article.
-
The impact of kidney function on Alzheimer's disease blood biomarkers: implications for predicting amyloid-β positivity.Alzheimers Res Ther. 2025 Feb 19;17(1):48. doi: 10.1186/s13195-025-01692-z. Alzheimers Res Ther. 2025. PMID: 39972340 Free PMC article.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
Cited by
-
Platycodin D and voluntary running synergistically ameliorate memory deficits in 5 × FAD mice via mediating neuromodulation and neuroinflammation.Front Aging Neurosci. 2024 Sep 25;16:1451766. doi: 10.3389/fnagi.2024.1451766. eCollection 2024. Front Aging Neurosci. 2024. PMID: 39385832 Free PMC article.
-
Plasma S100β is a predictor for pathology and cognitive decline in Alzheimer's disease.Fluids Barriers CNS. 2025 Jan 9;22(1):4. doi: 10.1186/s12987-024-00615-8. Fluids Barriers CNS. 2025. PMID: 39789614 Free PMC article.
-
A Positive Allosteric Modulator of the SERCA Pump Rescues Hippocampal Neuronal Circuits Dysfunction and Cognitive Defects in a Mouse Model of Alzheimer's Disease.J Neurosci. 2025 Jul 23;45(30):e2337242025. doi: 10.1523/JNEUROSCI.2337-24.2025. J Neurosci. 2025. PMID: 40555519
-
A novel miR-4536-3p inhibition ameliorates Alzheimer's disease by reducing Aβ accumulation and tau phosphorylation.Alzheimers Res Ther. 2025 Aug 12;17(1):189. doi: 10.1186/s13195-025-01834-3. Alzheimers Res Ther. 2025. PMID: 40796907 Free PMC article.
References
-
- Musaeus CS, Gleerup HS, Hasselbalch SG, Waldemar G, Simonsen AH. Progression of blood-brain barrier leakage in patients with Alzheimer's disease as measured with the cerebrospinal fluid/plasma albumin ratio over time. J Alzheimers Dis Rep. 2023;7(1):535–541. doi: 10.3233/ADR-230016. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

