Current Status of Indole-Derived Marine Natural Products: Synthetic Approaches and Therapeutic Applications
- PMID: 38535467
- PMCID: PMC10971852
- DOI: 10.3390/md22030126
Current Status of Indole-Derived Marine Natural Products: Synthetic Approaches and Therapeutic Applications
Abstract
Indole is a versatile pharmacophore widely distributed in bioactive natural products. This privileged scaffold has been found in a variety of molecules isolated from marine organisms such as algae and sponges. Among these, indole alkaloids represent one of the biggest, most promising family of compounds, having shown a wide range of pharmacological properties including anti-inflammatory, antiviral, and anticancer activities. The aim of this review is to show the current scenario of marine indole alkaloid derivatives, covering not only the most common chemical structures but also their promising therapeutic applications as well as the new general synthetic routes developed during the last years.
Keywords: biological activity; indole alkaloids; marine resources; synthetic strategies; therapeutic application.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures





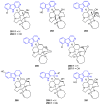
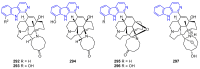
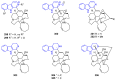
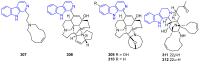
Similar articles
-
3-Acetyl Indole in the Synthesis of Natural Bioactive Compounds.Curr Org Synth. 2025;22(3):328-341. doi: 10.2174/0115701794325027240827043203. Curr Org Synth. 2025. PMID: 40259586 Review.
-
Marine-Derived Indole Alkaloids and Their Biological and Pharmacological Activities.Mar Drugs. 2021 Dec 21;20(1):3. doi: 10.3390/md20010003. Mar Drugs. 2021. PMID: 35049859 Free PMC article. Review.
-
Cyclohepta[b]indoles: A Privileged Structure Motif in Natural Products and Drug Design.Acc Chem Res. 2016 Nov 15;49(11):2390-2402. doi: 10.1021/acs.accounts.6b00265. Epub 2016 Oct 6. Acc Chem Res. 2016. PMID: 27709885
-
Chemistry and biology of fascaplysin, a potent marine-derived CDK-4 inhibitor.Mini Rev Med Chem. 2012 Jun;12(7):650-64. doi: 10.2174/138955712800626719. Mini Rev Med Chem. 2012. PMID: 22512549 Review.
-
Tryptophan derived natural marine alkaloids and synthetic derivatives as promising antimicrobial agents.Eur J Med Chem. 2021 Jan 1;209:112945. doi: 10.1016/j.ejmech.2020.112945. Epub 2020 Oct 21. Eur J Med Chem. 2021. PMID: 33153766 Review.
Cited by
-
3-Acetyl Indole in the Synthesis of Natural Bioactive Compounds.Curr Org Synth. 2025;22(3):328-341. doi: 10.2174/0115701794325027240827043203. Curr Org Synth. 2025. PMID: 40259586 Review.
-
Regio- and Enantioselective N-Heterocyclic Carbene-Catalyzed Annulation of Aminoindoles Initiated by Friedel-Crafts Alkylation.Org Lett. 2024 Aug 23;26(33):6993-6998. doi: 10.1021/acs.orglett.4c02434. Epub 2024 Aug 8. Org Lett. 2024. PMID: 39115978 Free PMC article.
-
Metabolomic and genomic insights into Micromonospora carbonacea subsp. caeruleus for anti-colorectal compound.Appl Microbiol Biotechnol. 2025 Feb 25;109(1):50. doi: 10.1007/s00253-025-13427-z. Appl Microbiol Biotechnol. 2025. PMID: 39998653 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

