Environmental Toxin Biliatresone-Induced Biliary Atresia-like Abnormal Cilia and Bile Duct Cell Development of Human Liver Organoids
- PMID: 38535810
- PMCID: PMC10974618
- DOI: 10.3390/toxins16030144
Environmental Toxin Biliatresone-Induced Biliary Atresia-like Abnormal Cilia and Bile Duct Cell Development of Human Liver Organoids
Abstract
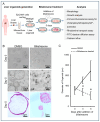
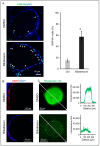
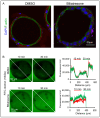
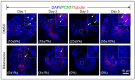
Biliary atresia (BA) is a poorly understood and devastating obstructive bile duct disease of newborns. Biliatresone, a plant toxin, causes BA-like syndrome in some animals, but its relevance in humans is unknown. To validate the hypothesis that biliatresone exposure is a plausible BA disease mechanism in humans, we treated normal human liver organoids with biliatresone and addressed its adverse effects on organoid development, functions and cellular organization. The control organoids (without biliatresone) were well expanded and much bigger than biliatresone-treated organoids. Expression of the cholangiocyte marker CK19 was reduced, while the hepatocyte marker HFN4A was significantly elevated in biliatresone-treated organoids. ZO-1 (a tight junction marker) immunoreactivity was localized at the apical intercellular junctions in control organoids, while it was markedly reduced in biliatresone-treated organoids. Cytoskeleton F-actin was localized at the apical surface of the control organoids, but it was ectopically expressed at the apical and basal sides in biliatresone-treated organoids. Cholangiocytes of control organoids possess primary cilia and elicit cilia mechanosensory function. The number of ciliated cholangiocytes was reduced, and cilia mechanosensory function was hampered in biliatresone-treated organoids. In conclusion, biliatresone induces morphological and developmental changes in human liver organoids resembling those of our previously reported BA organoids, suggesting that environmental toxins could contribute to BA pathogenesis.
Keywords: biliary atresia; biliatresone; organoids.
Conflict of interest statement
All authors declare no support from any organization for the submitted work, no financial relationships with any organizations that might have had an interest in the submitted work in the previous three years, and no other relationships or activities that could appear to have influenced the submitted work.
Figures

Similar articles
-
The toxin biliatresone causes mouse extrahepatic cholangiocyte damage and fibrosis through decreased glutathione and SOX17.Hepatology. 2016 Sep;64(3):880-93. doi: 10.1002/hep.28599. Epub 2016 May 20. Hepatology. 2016. PMID: 27081925 Free PMC article.
-
Impaired Redox and Protein Homeostasis as Risk Factors and Therapeutic Targets in Toxin-Induced Biliary Atresia.Gastroenterology. 2020 Sep;159(3):1068-1084.e2. doi: 10.1053/j.gastro.2020.05.080. Epub 2020 Jun 4. Gastroenterology. 2020. PMID: 32505743 Free PMC article.
-
Beta-amyloid deposition around hepatic bile ducts is a novel pathobiological and diagnostic feature of biliary atresia.J Hepatol. 2020 Dec;73(6):1391-1403. doi: 10.1016/j.jhep.2020.06.012. Epub 2020 Jun 16. J Hepatol. 2020. PMID: 32553668
-
Biliatresone: progress in biliary atresia study.World J Pediatr. 2023 May;19(5):417-424. doi: 10.1007/s12519-022-00619-0. Epub 2022 Sep 27. World J Pediatr. 2023. PMID: 36166189 Free PMC article. Review.
-
Biliary atresia: From Australia to the zebrafish.J Pediatr Surg. 2016 Feb;51(2):200-5. doi: 10.1016/j.jpedsurg.2015.10.058. Epub 2015 Nov 4. J Pediatr Surg. 2016. PMID: 26653951 Review.
Cited by
-
Generation of human induced pluripotent stem cell lines derived from patients of cystic biliary atresia.Hum Cell. 2024 Nov 13;38(1):18. doi: 10.1007/s13577-024-01147-x. Hum Cell. 2024. PMID: 39532815 Free PMC article.
-
Organoids in biliary atresia.World J Pediatr Surg. 2025 May 15;8(2):e001010. doi: 10.1136/wjps-2025-001010. eCollection 2025. World J Pediatr Surg. 2025. PMID: 40385243 Free PMC article. Review.
-
Genetic background and biliary atresia.World J Pediatr Surg. 2025 Jun 6;8(3):e001023. doi: 10.1136/wjps-2025-001023. eCollection 2025. World J Pediatr Surg. 2025. PMID: 40519535 Free PMC article. Review.
-
Prompt distinction of Alagille syndrome and biliary Atresia in infants: a comparative study.BMC Pediatr. 2025 Jul 9;25(1):543. doi: 10.1186/s12887-025-05896-y. BMC Pediatr. 2025. PMID: 40634917 Free PMC article.
-
Biliary atresia.Nat Rev Dis Primers. 2024 Jul 11;10(1):47. doi: 10.1038/s41572-024-00533-x. Nat Rev Dis Primers. 2024. PMID: 38992031 Free PMC article. Review.
References
-
- Jain V., Burford C., Alexander E.C., Sutton H., Dhawan A., Joshi D., Davenport M., Heaton N., Hadzic N., Samyn M. Prognostic markers at adolescence in patients requiring liver transplantation for biliary atresia in adulthood. J. Hepatol. 2019;71:71–77. doi: 10.1016/j.jhep.2019.03.005. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

