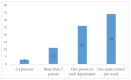
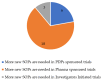
Operational Differences between Product Development Partnership, Pharmaceutical Industry, and Investigator Initiated Clinical Trials
- PMID: 38535880
- PMCID: PMC10975146
- DOI: 10.3390/tropicalmed9030056
Operational Differences between Product Development Partnership, Pharmaceutical Industry, and Investigator Initiated Clinical Trials
Abstract
Medicine development is a lengthy endeavour. Increasing regulatory stringency and trial complexity might lead to reduced efficiency, dwindled output, and elevated costs. However, alternative models are possible. We compared the operational differences between pharmaceutical industry sponsored trials, product development partnership trials, and investigator-initiated trials to identify key drivers of inefficiency in clinical research. We conducted an exploratory mixed-methods study with stakeholders, including clinical trial sponsors, contract research organisations, and investigators. The qualitative component included 40 semi-structured interviews, document reviews of 12 studies and observations through work shadowing in research institutions in Burkina Faso, Mali, and Switzerland. The findings were triangulated with an online survey polling clinical research professionals. The operational differences were grouped under five categories: (i) trial start-up differences including governance and management structure; (ii) study complexity; (iii) site structural and organisational differences; (iv) study conduct, quality approaches, and standard operating procedures; and (v) site capacity strengthening and collaboration. Early involvement of sites in the planning and tailored quality approaches were considered critical for clinical operations performance. Differences between the types of trials reviewed pertained to planning, operational complexities, quality approaches, and support to the sites. Integration of quality-by-design components has the potential to alleviate unnecessary process burden.
Keywords: academia; clinical trial operations; efficiency; pharmaceutical industry; product development partnership.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
The future of Cochrane Neonatal.Early Hum Dev. 2020 Nov;150:105191. doi: 10.1016/j.earlhumdev.2020.105191. Epub 2020 Sep 12. Early Hum Dev. 2020. PMID: 33036834
-
Understanding the investigators: a qualitative study investigating the barriers and enablers to the implementation of local investigator-initiated clinical trials in Ethiopia.BMJ Open. 2013 Nov 27;3(11):e003616. doi: 10.1136/bmjopen-2013-003616. BMJ Open. 2013. PMID: 24285629 Free PMC article.
-
Regulatory and operational challenges in conducting Asian International Academic Trial for expanding the indications of cancer drugs.Clin Transl Sci. 2021 May;14(3):1015-1025. doi: 10.1111/cts.12965. Epub 2021 Mar 1. Clin Transl Sci. 2021. PMID: 33382914 Free PMC article.
-
[Challenges in the organization of investigator initiated trials: in transplantation medicine].Chirurg. 2011 Mar;82(3):249-54. doi: 10.1007/s00104-010-1997-0. Chirurg. 2011. PMID: 21416397 Review. German.
-
Towards achieving interorganisational collaboration between health-care providers: a realist evidence synthesis.Health Soc Care Deliv Res. 2023 Jun;11(6):1-130. doi: 10.3310/KPLT1423. Health Soc Care Deliv Res. 2023. PMID: 37469292
Cited by
-
A qualitative study on the management system for investigator-initiated studies in healthcare institutions in Beijing, China.Health Res Policy Syst. 2024 Dec 18;22(1):165. doi: 10.1186/s12961-024-01264-x. Health Res Policy Syst. 2024. PMID: 39695849 Free PMC article.
References
-
- STCO Guidelines for Risk-based monitoring. [(accessed on 13 October 2023)]. Available online: https://www.sctoplatforms.ch/en/publications/guidelines-and-reports/guid....
Grants and funding
LinkOut - more resources
Full Text Sources